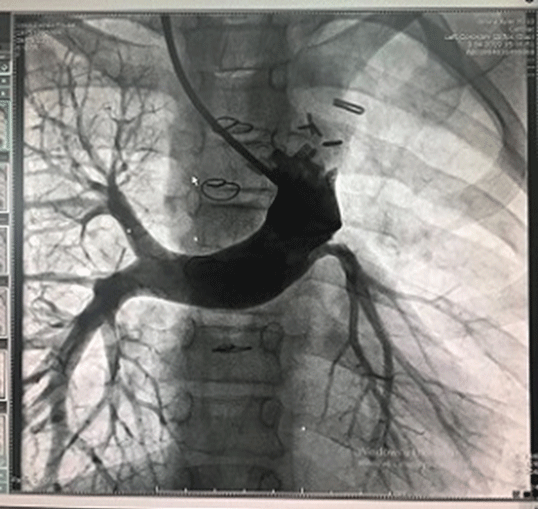
Figure 1: Hypoplastic left pulmonary artery.
Farid Gojayev1* Ruya Cholak2 Emin Tireli3
1Department of Cardiovascular Surgery, Bağcılar Training and Research Hospital; Istanbul, Turkey*Corresponding author: Farid Gojayev, Department of Cardiovascular Surgery, Bağcılar Training and Research Hospital; Istanbul, Turkey; E-mail: farid.gojayev@yahoo.com
Introduction: In this study, we present our experiences and results of the first Fontan surgeries we performed in our center in accordance with the surgical criteria we believe in.
Materials and methods: This is an observational study involving 19 patients. We investigated the data and survival rate of 19 patients who underwent Fontan surgery one by one. We wanted to know how the surgical method we used and the age factor affected our results. Our main hypothesis was to keep the cardiopulmonary bypass time very short and not to prefer the total circulatory arrest method.
Results: Patients were not homogeneous in terms of demographic characteristics. Patients differed in terms of pre- and postoperative cardiac diagnosis and complications. We lost 2 of 19 patients and the overall mortality rate was 10.5%. We used Extracardiac Membrane Oxygenation (ECMO) support in 3 patients. We applied Implantable Cardioverter Defibrillator (ICD) implantation to the patient. The discharge due to effusion due to chylothorax was prolonged in 4 patients. The postoperative NHYA capacity of the patients was evaluated as 1 or 2.
Discussion: We recommend that this operation should not be performed at a very young age, if the oxygen saturation value of heart patients who are candidates for Fontan is not very critical, if the ventricular ejection fraction capacity is not low, if the physical exertion capacity and examination findings are within normal limits.
Conclusion: The Fontan procedure is still the best long-term palliative operation for all single ventricle cases. We believe that it would be beneficial to postpone this surgery to the advanced age group as much as possible in order not to increase the mortality rate.
Fontan procedure; Congenital heart desease; Univentricular heart
When performing the surgical treatment of univentricular heart defects, there is a need to create a well-functional single ventricle that will provide balanced circulation to both the lungs and the body. The last step in the surgical correction of univentricular morphological heart defects is the Fontan procedure [1]. The main purpose of this surgery is to direct the circulation of the entire venous system directly to the pulmonary system without contacting the heart and to reduce the excess heart load. Old modification of classical Fontain surgery first performed in 1971 [2]. Until then, many patients born with a single ventricle would usually die within the first year of life. The aim of this multi-stage therapy is to reduce the volume load of the single ventricle by separating the pulmonary circulation from the systemic circulation, and to keep arterial blood saturation values close to normal by balancing the ratio (Qp/Qs) between pulmonary and systemic flow [3]. Thus, we can improve the long-term life span and quality. According to the latest published data, the 30-year survival expected to be approximately 89% (25) [4].
We performed a total of 279 congenital heart surgeries between 2018-2020. Of these, 19(6.81%) patients reached the Fontan procedure stage and we performed Fontan surgery on these patients. This was an observational study. We investigated the data and survival rate of 19 patients who underwent Fontan surgery one by one. We wanted to know how the surgical method we used and the age factor affected our results. Our main hypothesis was to keep the cardiopulmonary bypass time very short and not to prefer the total circulatory arrest method. Eleven of the patients were male and eight were female. According to the age limitation, 4 people were under 4 years old and 15 people were between 4-8 years old. The patient distribution according to primary cardiac anomalies was 1 Ebstein anomaly, 7 Tricuspid Atresia (TA), 2 tricuspid atresia+Pulmonary Stenosis (PS), 5 Hypoplastic Left Heart Syndrome (HLHS), 2 double-inlet left ventricle, 2 transposition of the great arteries (TGA)+PS. Additional pathologies were Atrial Septal Defect (ASD), Ventricular Septal Defect (VSD). The classification of the patients according to their primary diagnosis is shown in table 1. Left pulmonary artery of a patient with TGA+PS was critically hypoplasic. We completed the Fontan circulation in this patient with the existing unilateral right pulmonary artery (Figure 1). BlalockTaussig shunt had been applied to three patients previously to improve pulmonary blood circulation. In a study on the complaints of edema, low saturation and bruising in a TA+PS patient 3 months after Glenn shunt surgery, stenosis in the anastomosis line was found. There upon, we applied balloon to the stenotic area and stenting to the right ventricular outflow tract to improve antegrade pulmonary flow (Figure 2). Upon stabilization of the patient’s condition, we underwent an elective Fontan operation. The application complaints of all cases consisted of findings such as bruising, shortness of breath, tachypnea, fatigue, decreased effort capacity and palpitation growth and development retardation, and heart failure.

Figure 1: Hypoplastic left pulmonary artery.
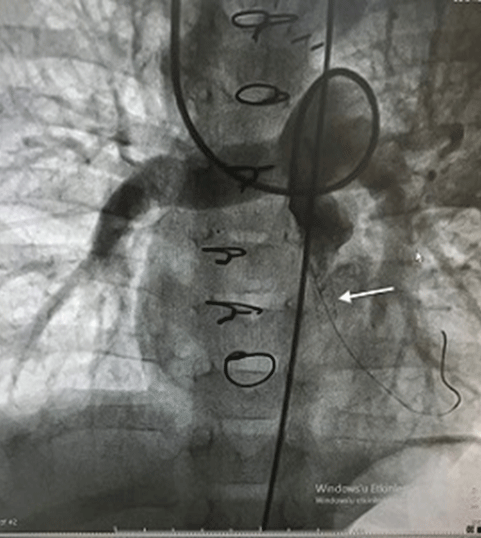
Figure 2: Stent placed in the right ventricular outflow tract and balloon dilatation of Glenn’s anastomosis.
| Total Patients | TA | TA+PS | DILV | TGA+PS | HLHS | Ebstein Anomaly |
| 19 | 7 | 2 | 2 | 2 | 5 | 1 |
Table 1: Distribution of patients included in the study according to their primary diagnosis.
*TA: Tricuspid Atresia
*PS: Pulmonary Stenosis
*BAT: Great Artery Transposition
*HLHS: Hypoplastic Left Heart Syndrome
*DİLV: Double-Inlet Left Ventricle
Antegrade aortic cannulation was performed with a median sternotomy under general anesthesia. Selective cannulation of superior and inferior vena cava was performed in all patients. Mild hypothermia 28-32°C was achieved under Continuous Cardiopulmonary Bypass (CPB). During the cooling, we completed the adherent tissue dissections. Particularly, regions with continuity of pulmonary arteries located behind the ascending aorta were liberated. We used only one dose of hot blood cardioplegia (30 ml/kg) through the aortic antegrade route during the entire operation. We took care to keep the cross-clamp time short (13.2 ± 8.7 min). We did not prefer the total circulatory arrest method in any patient. All cases had previously undergone a bidirectional Glenn shunt operation. We did not use a patch for tricuspid valve closure between the right atrium and ventricle in our cases. We preferred the extra cardiac tunnel method using 16-20 size gore-tex tube grafts in all cases. The graft was anastomosed endto-end to the right pulmonary artery, and to the vena cava inferior, 1 cm above the diaphragm. The graft anastomosis was performed endto-end by transecting 1 cm above the diaphragm into the vena cava inferior. In the right pulmonary artery, it was performed medial to the anastomosis of the glans by the end-to-side method. In the right pulmonary artery, it was applied to the medial side by the end-side method. We routinely fenestrated a 5 mm diameter between the goretex graft and the right atrium. At the end of the operation, no graft deformation was observed and the oxygen saturation value increased over 95% (Figure 3). Our study was approved by the ethics committee decision dated 01.06.2021 and numbered 2021/11. The study was conducted in accordance with the Principles of the Declaration of Helsinki.
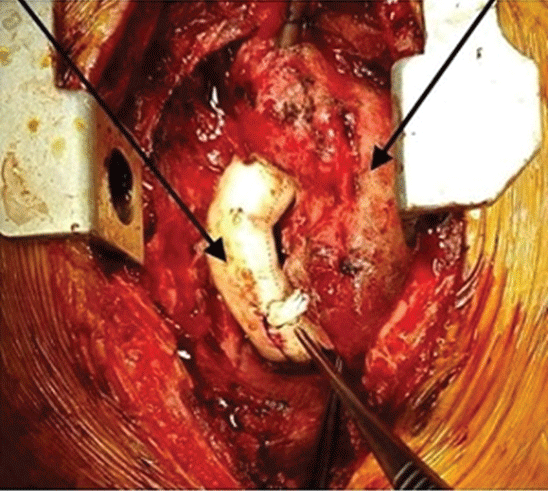
Figure 3: Our Fenestrated extracardiac type Fontan operation.
Preoperative electrocardiogram (ECG) was sinus rhythm preoperatively in all patients. In echocardiographic findings, mean pulmonary artery pressures were 10(5-18) mmHg and mean pulmonary vascular resistance was 1.9 ± 0.7 Wood. The mean value of functional single ventricular ejection fraction before surgery was 59.4 ± 9.0%. Four patients had mild to moderate atrioventricular valve insufficiency, and three of them were in the under-4 age group. Other preoperative data of the patients are shown in table 2. Eleven patients were extubated on the first postoperative day and 6 patients were extubated within 2-3 days. All patients were given acetylsalicylic acid (100 mg/day) as antiaggregant prophylaxis after discharge. When we looked at the mortality rates of the cases, we lost 2 of 19 patients and the overall mortality rate was 10.5%. One of these cases was 38 months old and the other was 3 years old. We lost one of the patients as a result of acute heart failure that developed at the time of pump discharge, and the other due to low cardiac output during the intensive care period in the first 72 hours postoperatively. We used Extra Cardiac Membrane Oxygenation (ECMO) support in 3 patients. Supraventricular arrhythmia occurred in two patient in the early postoperative period. First of all, antiarrhythmic medical therapy was started. While one of these patients responded positively to medical treatment, the other could not be successful. Sinus rhythm was not achieved despite medical treatment. The cardioversion procedure that followed was successful, hemodynamically stable. However, we applied ICD implantation to this patient after 2 months due to intermittent extrasystolic beats. 7 patients were extubated within the first 24 hours and 8 patients within the first 48 hours. We encountered prolonged extubation in 2 patients, that is, in the first 3 days. While 13 patients could be discharged within 7-12 days, discharge in 4 patients varied between 15-35 days due to effusion due to prolonged chylothorax. There were no patients who needed hemodialysis with renal dysfunction. No mortality was observed in the near term after discharge. Outpatient follow-up of all patients was done at 3-month intervals. After discharge, the first echocardiography and other controls were performed 1 month later at 3-month intervals. The patients were followed for 1-3 years, with a mean of 17.2 ± 8.4 months. One year after the operation, signs of peripheral edema were observed in the areas around the eyes and in the lower extremities in 2 patients (Figure 4). Protein malnutrition was diagnosed in laboratory tests. In these patients, no significant gradients were detected in the anastomosis areas in the control angiography (Figure 5). One patient had partial graft thrombosis 3 months after discharge. The patient was hospitalized and a 1-week medical treatment was performed. The patient was discharged as the symptoms regressed. In addition, cardiac functions were seen well on echocardiography. In the postoperative 1-2 year echocardiographic follow-up of all patients, the functional single ventricle ejection fraction value was measured as 63.4 ± 10% (p=0.085). NYHA capacity of the patients was evaluated as 1 or 2.3Postoperative data of the patients are shown in table 3.
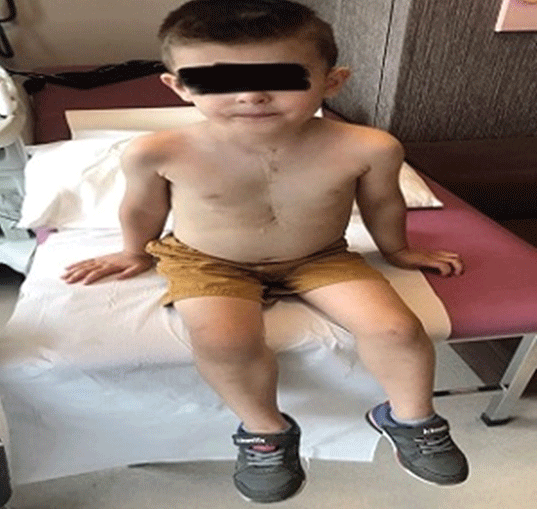
Figure 4: Edema findings around the eyes and in the body after 1 year in the patient who underwent Fontan surgery.
| Ventricular morphology Dominant left ventricle Dominant right ventricle Biventricular |
12 5 2 |
| Preoperative EF | 59.4 ± 9.0 |
| Elevated creatinine | 6 |
| Dialysis | 0 |
| Pleural effusion | 3 |
| Systemic AVV regurgitation | 4 |
| Previous interventions (Branch PA/PDA stenting) | 14 |
| Cirrhosis | 0 |
| Prior stroke | 0 |
| Atrial arrhythmias | 0 |
| Permanent pacemaker | 1 |
Table 2: Preoperative baseline evaluation.
*EF: Ejection Fraction
*AVV: Atrioventricular Valve
*PA: Pulmonary Artery
*PDA: Patent Ductus Arteriosus
| Discharge (In the 7-35 days) | In 7-35 days 17 |
| Postoperative EF | % 63.4 ± 10 (p=0.085) |
| Cerebrovascular accident | 0 |
| Arrhythmias | 2 |
| Protein-losing enteropathy | 2 |
| Prolonged drainage (chylothorax) | 4 |
| Extubation (In the first 3 days) | 17 |
| Bleeding revision | 1 |
| Fontan graft thrombosis | 1 |
| Using ECMO | 1 Successful; 1 Unsuccessful |
| ICD implantation | 1 |
| NHYA capacity | 1-2 |
Table 3: Postoperative evaluation.
EF: Ejection Fraction
ECMO: Extra Cardiac Membrane Oxygenation
ICD: Intracardiac Defibrillator
NHYA: New York Heart Association
In 1971, the first fontan surgery was performed by Francis Fontan and Eugene Bauder. Although this surgery was originally described for the treatment of tricuspid atresia, it was later chosen as the complementary stage of all univentricular pathologies [2]. In the 1970s, when it was first applied, mortality rates were between 20-35% [5]. In the 2000s, this rate decreased to around 7-10% with detailed research of some risk factors and various measurements. In the late 1970s, Choussat, et al. Published criteria that included the applicability of the Fontan procedure to which patients. After this period, the cases were evaluated according to the preoperative anatomical and physiological Fontan criteria. Decisions to wait or other palliative operations were made for cases that did not meet the criteria. One of the most important of these criteria is Pulmonary Vascular Resistance (PVR). Normal PVR index should be <1.5 Wood unit. When the PVR index is >3 Wood units, the presence of pulmonary hypertension (PH) can be mentioned. In cases that are candidates for Fontan surgery, the desired limit is a maximum of 2-3 wood/unit [6]. Vasodilator application is sometimes recommended for patients with a basal PVR index between 4 and 6 Wood units. Some patients may have a 20% reduction in the PVR index with this. Another important factor is the mean Pulmonary Artery Pressure value (PAP). Normal value of PAP is 15-18 mmHg. In cases that are candidates for Fontan surgery, the average PAP should be <15 mmHg [7]. Normally developed right and left pulmonary arteries, functional single ventricle with good ejection fraction, absence of atrioventricular valve insufficiency, perfect venous return and normal sinus rhythm are other criteria for performing Fontan surgery [8]. The effect of age on mortality is also important. Likewise, both cases we lost were in the low age group. In the results of our clinic, the overall operative mortality rate can be seen as high as 10.5%. These two patients, especially under the age of 4, have contributed to this rate. On the other hand, there was no mortality in our patients over the age of 4 (17 patients). The mechanism by which the patient’s age affects mortality is related to cardiovascular anatomy and pulmonary vascular structure reactivity that have not completed their development [9]. Among the causes of mortality in patients who have undergone Fontan surgery, arrhythmia, thromboembolism, cerebrovascular events and systemic venous return tract narrowing are also included [10]. As we know, there are 2 types of Fontan surgery as lateral tunnel and extra cardiac type. Both methods have different features. We prefer the extra cardiac type modification in all our cases. Considering that arrhythmia and narrowing of the venous return tract are more likely in the lateral type method, these risks are less in extra cardiac type fontan [11]. On the other hand, the extracardiac type is more advantageous in cases where the systemic venous connection is on the same side in the malposition of the apex of the heart. Using an excessively large size graft compared to the patient’s IVC size may cause hemodynamic disturbance. We know that the IVC in an adult patient is approximately 20 mm. For children, this size is formed when they reach the age of about 15 kg and 3 years old. But sometimes, although body weight reaches the desired values, the development of cardiovascular structures is not the same in parallel. In this case, we think that it is correct to choose the conduit size based on the intraoperative evaluation of the actual IVC size. Our preference for the use of conduit was to apply one above the IVC size that we measured intra-operatively, especially in 3-year-old cases. Generally, there is an opinion that 16-18 mm conduits are ideal for 3-3,5 year old children [12]. Lardo AC, et al. Alexi-Meskishvili VA, et al. have important ideas in their conduit-IVC size selection. In cases where 20 mm and larger conduits were used at this age, stagnation in excess fluid volume in the lateral graft and backflow in the lateral part during expiration was shown. There are still various controversial opinions regarding the opposition or shifting of the superior vena cava with conduit anastomosis in cases with Glenn shunt. Our preference is to place the conduit anastomosis in a more medial area. With this method, we consider a more even and better distribution of blood to the right and left lungs [13,14]. Some patients may have difficulties in keeping up with the new physiology after the fontan operation. The most important of these is prolonged pleural effusions. Especially chylothorax, protein-losing enteropathy and some lung problems. We discharged a total of 4 patients late due to prolonged chylothorax consistency drainage. Appropriate diet was used in the treatment of chylothorax. We supported patients’ intake of adequate calories and protein while restricting their consumption of fat containing mediumchain triglycerides [15]. Chronic passive stasis may occur in the venous system and liver after Fontan surgery. Secondary inflammation and increased platelet activity and thromboembolic complications may occur. This can be considered an unwanted disadvantage [16]. Stroke is one of the most important complications in patients who have undergone Fontan surgery [17]. We have not encountered such events in our series. Anticoagulation and antiaggregation use in different combinations is required against hromboembolism. In all patients, we routinely preferred low molecular weight heparin for the first 48 hours postoperatively and then aspirin tablet 100 mg/day for 6 months. We did not encounter any thrombosis event in the short and medium term impression. In this regard, the choice of treatment protocol is subjectively different in separate centers. After thromboembolic events develop, the mortality rate can be 38% despite aggressive treatment [18]. When we compared the functional single ventricle load before and after surgery, it was more spherical, the muscle mass was partially hypertrophic and the volume was higher before the operation. Positive changes were reflected in these parameters after the operation. The diastolic functions of the ventricle are important, especially in providing pulmonary circulation. Sometimes, after the fontan procedure, ventricular diastolic function deteriorates secondary to relaxation disorder, and this slows down the systemic venous circulation [19]. Throughout our study, we did not follow any patient with ventricular systolic and diastolic dysfunction, except for 2 patients we lost. Previously known myocardial dysfunction or atrioventricular valve insufficiency was not observed in the patients who were lost. In a 10-year study by Mira Fauziah et al. [20] systemic ventricular morphology was not associated with the 10-year survival rate after Fontan surgery. The probable cause was the problem of meeting the systemic venous return by the pulmonary system, diameter mismatch or increased pulmonary vascular resistance. Today, Fontan surgery is usually performed by applying fenestration [21]. In our case series, we applied 3-5 mm of routine fenestration in all cases. Fenestration slightly increases cardiopulmonary bypass time, saturation value may be slightly lower in the early period, but decreases the incidence of arrhythmia [22]. We believe that fenestration reduces the incidence of postoperative effusion in the short term and shortens the length of hospital stay. Fenestration helps reduce the pressure of the inferior vena cava. It usually closes on its own in the long term. In the angiography we performed for 1 year sonar control, we saw that the fenestration was closed (Figure 5). Some centers also perform Fontan surgery without the use of fenestration. In most studies, approximately 70% of these fenestrations closed spontaneously. Fontan surgery can be performed easily as long as it meets all the surgical indication conditions mentioned above. Here, along with other indications, the main goal is the systemic venous circulation to be included in the pulmonary system without interfering with the heart and to ensure circulation [23-26]. This procedure has the hemodynamic advantages mentioned above and is still a good palliative operation for single ventricle cases.
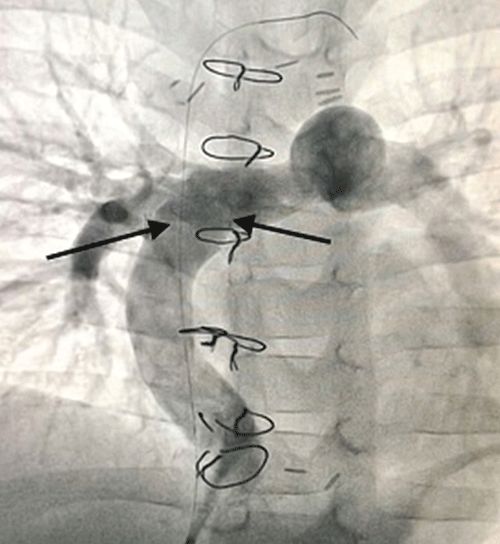
Figure 5: Control angiography 1 year after surgery, no stenosis in fontan anastomosis and fenestration spontaneously closed.
In order to reduce mortality and morbidity, the result we have inferred from our case series is to move the age factor to more advanced ages as possible. We have the opportunity to say this, especially since the patients we lost are under the age of 4. If the preoperative oxygen saturation value of the patients is not critical, if the functional single ventricle function is normal, if there is no significant leakage in the atrioventricular valve, if the physical exertion capacity and hemodynamic circulation findings are close to normal, it is useful to wait as long as possible between the Glenn shunt and Fontan surgery. In conclusion, we think that the morbidity and mortality risk of surgery performed at an older age is low.
There is no conflict of interest in the preparation and publication of this article.
There is no financial support for the research and authorship process of this article.
All authors read and approved the final version of the manuscript.
Download Provisional PDF Here
Article Type: RESEARCH ARTICLE
Citation: Gojayev F, Cholak R, Tireli E (2022) Fontan Operation Experience and Results of our Cardiovascular Surgery Center. J Surg Open Access 8(3): dx.doi.org/10.16966/2470-0991.267
Copyright: © 2022 Gojayev F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access