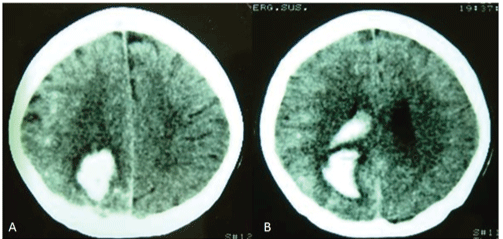
Figure 1: Patient 1, a 17-year-old female with sudden headache. CT scan shows brain hemorrhage with ventricular drainage (A and B).


Luciano Bambini Manzato1* José Ricardo Vanzin2 Leonardo Frighetto3 Paulo Mesquita Filho4 Eduardo Pedrolo1 Marcos Paulo Tercziany Vanzin1 Alex Roman1
1Interventional Neuroradiology Department, Neurology and Neurosurgery Service, Passo Fundo, Rio Grande do Sul, Brazil*Corresponding author: Luciano Bambini Manzato, Interventional Neuroradiology Department, Neurology and Neurosurgery Service, Passo Fundo, Rio Grande do Sul, Brazil, Tel: +55 54 3317- 6022; E-mail: lbmanzato@hotmail.com
Multiple arteriovenous malformations (AVM) are relatively rare, accounting for 0.3 to 4.9% of all AVMs. We describe two cases of young patients with multiple cerebral AVMs and discuss their diagnosis and treatment planning. Both patients presented with sudden headache. In patient 1, a 17-year-old female, angiography demonstrated multiple right-sided AVMs. In patient 2, a 13-year-old male, head CT showed a right parietal intraparenchymal hemorrhage. Following emergency surgery to evacuate the hematoma, cerebral angiography demonstrated two separate nidi filled by the right posterior cerebral artery. Multiple cerebral AVMs are a rare and therapeutically challenging condition. While they are often syndromic, we were unable to diagnose any identifiable syndromes in our patients. A multidisciplinary approach is needed to discuss and decide the best treatment strategy for each patient. Treatment is as for single AVMs, and must take into account bleeding, location, size, and number of malformations.
Multiple arteriovenous malformation; Brain; Endovascular; Embolization
Cerebral arteriovenous malformations (AVMs) are lesions that occur due to abnormalities in development of the arteriovenous network. AVMs are characterized by a central nidus with feeding arteries and drainage veins, forming a low-resistance, high-flow shunt that dilates the arteries and thickens the vein walls. There is usually no neural tissue in the central nidus, which is the most likely site of bleeding. Multiple AVMs are relatively rare [1,2], occurring mostly in children and accounting for 0.3 to 4.9% of all such malformations [3-6]. However, the disease can cause significant long-term morbidity and mortality in the affected population. In this article, we describe two cases of patients with multiple cerebral AVMs, discuss a treatment strategy using liquid embolic agents, and address the role of stereotactic radiosurgery and microsurgery in managing this condition.
The first patient, a 17-year-old girl, presented with sudden headache. A head CT scan showed a right parietal intraparenchymal hemorrhage (Figure 1), and cerebral angiography revealed a right-sided multiple AVM (Figure 2) with multifocal niduses completely separated by normal brain tissue. Each AVM was completely independent, with afferent vessels and one superficial draining vein. Intranidal aneurysms were observed in all malformations.

Figure 1: Patient 1, a 17-year-old female with sudden headache. CT scan shows brain hemorrhage with ventricular drainage (A and B).
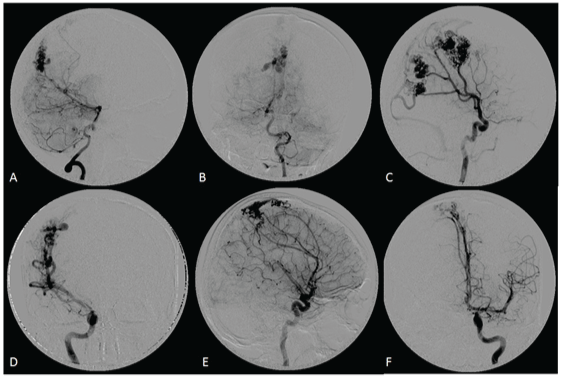
Figure 2: Cerebral angiogram showing multiple separate AVMs. Vertebral injection demonstrating midline AVM supplied by the right posterior cerebral artery (A, B). The middle cerebral artery supplies three separate nidi (C, D). Right anterior cerebral artery flow is provided only from the left carotid artery and feeds a parietal nidus (E, F).
Neurological examination demonstrated left homonymous hemianopsia. The treatment strategy was to first address the AVM that had bled, as determined by control MRI and CT. Clinical history and physical examination did not show any abnormalities suggestive of Hereditary Hemorrhagic Telangiectasia (HHT or Rendu-Osler-Weber disease), Wyburn-Mason syndrome, or other syndromes associated with cerebral AVMs. The patient was treated four times via endovascular approaches. Each session was performed by the same neuroradiologist, with the patient under general anesthesia. Systolic blood pressure during each procedure was controlled between 100 and 110 mmHg. Catheterization was performed via the transfemoral route with standard coaxial techniques. DMSO-compatible Ultra Flow 1.5F and Marathon 1.3F microcatheters (ev3, Irvine, CA, USA) were navigated to the nidus of the AVM, guided by a 0.010 or 0.008-inch guide wire. Once the microcatheter tip was in the desired position, Onyx® liquid embolic agent (ev3, Irvine, CA, USA) was injected. Briefly, the microcatheter was flushed with 0.25 mL of DMSO to fill the dead space; then, Onyx was aspirated into a 1-mL syringe, and 0.25 mL of this amount was injected slowly for 40 seconds to fill the microcatheter and replace the DMSO in the dead space; finally, slow injection of the liquid embolic agent was then continued under fluoroscopic guidance. Angiographic control was maintained during injections to assess nidus occlusion and the status of draining veins. When any reflux of Onyx into the feeding pedicle or venous migration was noted, the injection was stopped for 30-60 seconds to allow solidification, and then continued. Complete occlusion of each malformation was achieved after embolization (Figure 3).
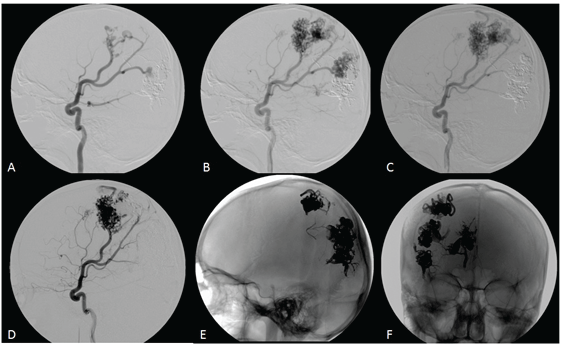
Figure 3: Multiple embolization sessions. First session with Onyx showing occlusion of the most inferior nidus (A). Second and third sessions with liquid embolic agent (B, C). Fourth procedure shows partial occlusion of a fronto-parietal nidus (D). Final appearance (E, F).
All procedures were performed under good technical conditions, and no clinical complications occurred. However, during the second procedure, the microcatheter was trapped at the end of treatment during the vertebral approach, and was thus left inside the vascular system, constituting a technical complication. The patient was discharged after 5 days. The interval between embolization sessions was approximately 3 months. As the last malformation was located in the motor area, an awake balloon occlusion test was performed. The patient did not tolerate the test, and developed left hemiparesis and hemiparesthesia; after a multidisciplinary discussion, the team’s decision was to refer the patient for radiosurgery.
The second patient, a 13-year-old male, presented with sudden headache and a Glasgow Coma Score of 15. Head CT showed a right parietal intraparenchymal hemorrhage (Figure 4). Due to deteriorating neurological status, an emergency craniotomy was performed to evacuate the hematoma. Subsequent angiography demonstrated right parietal and occipital AVMs with two separate nidi filled by the right posterior cerebral artery. As in Patient 1, each AVMs was completely independent, with afferent feeders and a single superficial draining vein (Figure 5).
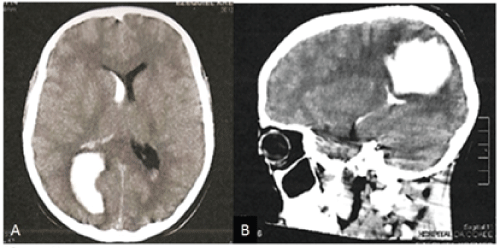
Figure 4: Patient 2, a 13-year-old male with severe headache. CT scan demonstrates parieto-occipital bleeding with ventricular drainage.
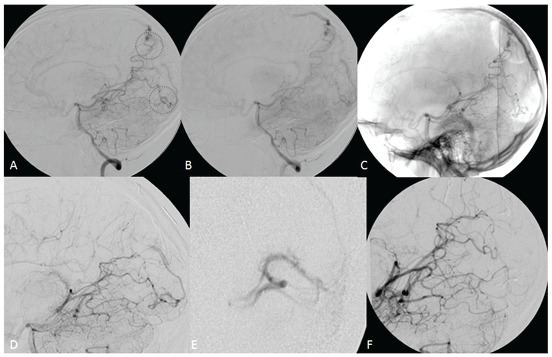
Figure 5: Two completely separated AVMs, both fed by the right posterior cerebral artery (A, B, C). Angiogram after surgical management of the parietal AVM (D). Selective injection of the occipital AVM before embolization (E). Final result with complete exclusion of both nidi.
A different treatment strategy was employed in this case. The right parietal AVM, identified as the bleeder lesion, was first resected completely via a microsurgical technique. This lesion was classified as Spetzler-Martin grade 1. No complications were reported during the procedure. On the same day, the patient underwent endovascular treatment of the second AVM. Angiography showed complete exclusion of this lesion (Figure 5).
The endovascular technique was largely similar to that employed in the first case, but biological glue was used. A Marathon 1.3F microcatheter (ev3, Irvine, CA, USA) was navigated into the nidus of the AVM with aid of a 0.010-inch guidewire. The microcatheter was flushed with 5 mL of 5% dextrose, and 20% Histoacryl® glue (B. Braun Melsungen AG) diluted in ethiodized oil (lipiodol®) was slowly injected under fluoroscopic guidance until complete angiographic exclusion of the nidus had been achieved (Figure 5). The patient was discharged 7 days after both procedures, with a Glasgow Outcome Score of 15.
Arteriovenous malformations are the most common vascular malformations to become symptomatic and require medical treatment. Multiple cerebral AVMs are rare [2,4], except when associated with HHT or unilateral retinocephalic vascular malformation [1,2]. They are most frequent in childhood, as in our case series. Although the large majority of cerebral AVMs probably occur congenitally due to failure of capillary formation during early embryogenesis [7], there is recent evidence that not all such AVMs are congenital in origin [8]; in fact, single AVMs are infrequent in childhood [5,9]. Some cerebral AVMs appear to form in response to a postnatal stimulus of angiogenesis, particularly in younger patients. Torné et al. [10] reported one case of multiple frontal cerebral AVMs possibly related to hypoplasia of the superior sagittal sinus.
Multiple cerebral AVMs normally have multifocal niduses completely separated by normal brain tissue. Angiography is essential for diagnosis of all AVMs, but multiple AVMs cannot be easily diagnosed by this method alone. A single nidus with multiple angiographic compartments should be distinguished from multiple niduses. The incidence of multiple AVMs is difficult to estimate accurately, but appears to range from 3% to 6% [2]. Multiple AVMs fall into two major categories: 1) simple or 2) complicated by known diseases [2]. The second category includes three subgroups: 1) HHT; 2) Wyburn-Mason syndrome or unilateral retinocephalic vascular malformation; and 3) soft-tissue vascular malformation. The incidence of hemorrhage is similar to that for single AVM, reported to be 2-3% [2,3], followed by convulsion and hydrocephalus. The most common location is the parietal lobe, followed by the frontal and temporal lobes [2].
The management of patients harboring multiple AVMs can be problematic. The literature on this condition is scarce, and there is no information regarding the risk of bleeding of each AVM alone [11]. Boone et al. [12], in a retrospective analysis of 21 patients under age 21, reported an annual risk of bleeding of 2.1%, similar to that of single AVMs. The principles of treatment for single AVM also apply to multiple AVMs. The treatment strategy should be based on a case-by-case evaluation and on hemodynamic characteristics. Robert et al. [6] described a case series of 6 adult patients harboring multiple brain AVMs. The authors suggest treatment of all present AVMs in case of bleeding, and treatment of unruptured AVMs only if angiographic risk factors are present (associated aneurysm, deep location, only deep venous drainage, and single draining vein). Lizuka Y et al. recommended intervention only for hemorrhagic AVM. According to other reports, remaining AVMs may bleed secondary to surgery [4]. In these cases, hemorrhage after resection of one AVM is attributed to hemodynamic stress [13]. Thus, unruptured AVMs should be addressed surgically as soon as possible after resection of a hemorrhagic AVM [4]. Considering the risk of hemorrhage and life expectancy at the typical age of detection, when patients are in good clinical condition, treatment of multiple cerebral AVMs should be performed as soon as possible. In our cases, there was no question as to whether treatment was indicated, because both patients presented with hemorrhage.
Several treatment modalities are used to manage AVM: microsurgical excision, endovascular embolization, stereotactic radiosurgery, or a combination thereof [14]. Various factors influence whether or how an AVM should be treated, including patient age and clinical condition, presentation, AVM location, size, morphology, complexity, expected natural history, and treatment risks. As noted above, management needs to be individualized to each patient, and careful study of all available imaging, including CT, MRI, and detailed cerebral angiography, is required. The ARUBA (A randomized trial of unruptured brain arteriovenous malformations) trial concluded that conservative treatment is safer than any intervention in patients with unruptured AVMs. However, it had many limitations, and we do not believe current management of the condition should change on the basis of this trial alone. Selected unruptured AVMs, with a low Spetzler-Martin grade and in non-eloquent areas, should be considered for treatment.
Yasargil described the treatment of multiple AVMs as essentially the same as for solitary lesions. Tada et al. emphasized that the size and the structures involved are more important factors in the prognosis than the multiplicity of the lesions. In multiple AVMs with cerebral hemorrhage, the bleeding lesion should be treated first [1].
Microsurgical AVM excision, which consists of removing the entire nidus of the AVM, is the most effective treatment currently available. However, not every AVM is amenable to or best treated with surgery. Grading systems, such as the Spetzler-Martin classification, are intended to predict the risk of neurologic deficit after surgery [14].
Many factors should be considered before endovascular treatment (EVT), i.e., embolization, of an AVM. This treatment modality was used primarily as an adjunct to surgery or radiosurgery to reduce blood flow and eliminate surgically inaccessible arterial feeders preoperatively, or to incrementally reduce the size of a large AVM nidus and make it amenable to surgical or radiosurgical treatment. Many reports have demonstrated the advancement and development of this technique. In the past, embolization with polyvinyl alcohol (PVA) particles, either alone or in combination with alcohol or coils, was used to facilitate surgical removal of these lesions. Alternatively, EVT using liquid adhesives, such as n-butyl cyanoacrylate (n-BCA), was used in numerous centers either for preoperative reduction of AVM size or for anatomic cure, which is achievable in only a limited number of patients [15]. The most substantial evolution in the treatment of cerebral AVMs was provided by the introduction of the Onyx® liquid embolic system. Onyx® is a biocompatible polymer containing ethyl vinyl alcohol dissolved in the organic solvent, dimethyl sulfoxide (DMSO). It is characterized by a different process of solidification that consists of copolymer precipitation instead of polymerization (i.e., as in n-BCA). This allows a slower, more controllable injection, and should result in more effective filling of the AVM nidus.
Stereotactic radiosurgery, in particular linear particle accelerator (LINAC) or Gamma Knife radiosurgery, can be used in select patients to provide a single, high dose of stereotactically localized radiation to the AVM nidus [14]. This radiation dose causes endothelial damage, smooth muscle cell proliferation, progressive sclerosis, and subsequent thrombosis of nidal channels over time. The success of stereotactic radiosurgery depends on AVM size and on the radiation dose delivered. Several clinical studies have demonstrated that AVM obliteration can be expected between 1 and 2 years after radiosurgery, provided the AVM nidus is less than 2–3 cm in diameter or has a volume of less than 10 mL [16-21]. It is not the treatment of choice in cases of ruptured AVM because of the time between irradiation and nidus occlusion.
In our first case, we achieved cure by the endovascular approach using Onyx® in three AVMs, without need for further surgery. Operative treatment was not our first choice because multiple craniotomies would have been required to access all AVMs; furthermore, one of the niduses (ultimately addressed by radiosurgery) was located on the motor gyrus. On the other hand, in Patient 2, the ruptured AVM was successfully addressed by microsurgery without any complications, while the other AVM was occluded with glue. This demonstrates the necessity of a multidisciplinary approach to patients with multiple AVMs.
Multiple cerebral AVMs are a rare and therapeutically challenging condition. A multidisciplinary team should discuss and decide the best treatment for each patient. Although such malformations are often syndromic, in our case series, we were unable to diagnose any established syndromes. Treatment approaches are similar to those for single AVMs, and must take into account bleeding, location, size, and number of malformations.
Download Provisional PDF Here
Article Type: Case Series
Citation: Manzato LB, Vanzin JR, Frighetto L, Filho PM, Pedrolo E, et al. (2017) Multiple Cerebral Arteriovenous Malformation: Report of Two Cases and Treatment Strategy. J Neurol Neurobiol 3(1): doi http://dx.doi.org/10.16966/2379-7150.138
Copyright: © 2017 Manzato LB, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access