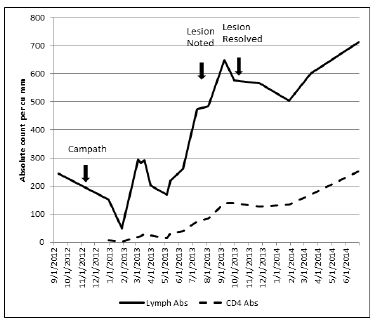
Figure 1: Absolute lymphocyte and CD4 cell count before and after Campath™ therapy.

Kevin Engelhardt1 Jane Siegel2 Bibhuti B Das1*
1Division of Cardiology, Department of Pediatrics, Children’s Medical Center, UTSW Medical Center, USA*Corresponding author: Bibhuti Das, Associate Professor, Division of Cardiology, Children’s Medical Center, UTSW Medical Center, 1935 Medical District Dr, Dallas, Texas 75235, USA, Tel: 214-456-2333; E-mail: bdas99@hotmail.com
Patients who have undergone organ transplantation can present with a variety of oral lesions that appear to be related either directly to their medication or arise as a consequence of drug-induced immunosuppression. Such lesions are commonly caused by fungal and viral agents. Campath™ (Alemtuzumab, a B and T cell depleting monoclonal antibody against CD-52) is used in cardiac transplant patients with refractory rejection. The profound T-cell suppression that occurs with Campath™ therapy broadens the differential diagnosis for such lesions to include potentially severe etiologies typically considered in patients with HIV. This report reviews the causes of oral lesions in immunosuppressed patients, specifically in the context of Campath™ therapy and describes an unusual case of a benign cause of a new oral lesion, chronic biting of the oral mucosa or Morsicato mucosae in a pediatric heart transplant recipient who was treated with Campath™ for cardiac allograft rejection.
Oral lesion; Heart transplant; Campath™ therapy; Immunosuppression
There are few case reports of the use of Alemtuzumab (Campath -1H; Berlex, Montville, NJ) in pediatric orthotopic heart transplantation [1]. However, this agent has been used in other solid organ transplant patients with refractory rejection, in hematopoietic stem cell transplant patients with refractory graft versus host disease (GVHD) and for conditioning, and in treatment of refractory hemophagocytic lymphohistiocytosis (HLH). The severe immunosuppressive effects of this therapy broaden the potential pool of opportunistic pathogens in the host. The effect of Campath™ is similar to the immunodeficiency state associated with acquired human immunodeficiency syndrome (AIDS) with increased vulnerability to opportunistic infections and malignant transformation. In this report, we present a case of refractory rejection requiring Campath™ therapy and the onset of new white oral mucosal lesions with a brief review of differential diagnosis of common white oral lesions in solid organ transplant patients.
An 11 year old female underwent her first orthotopic heart transplantation (OHT) at the age of 7 years for restrictive cardiomyopathy. She had done well for 4 years until she developed severe clinical rejection with hemodynamic compromise requiring placement of a percutaneous ventricular assist device, Impella™ (Abiomed, Danvers, MA). She underwent cardiac catheterization immediately after placement of the Impella™. Significant findings at catheterization were coronary artery vasculopathy (ISHLT grade I); endomyocardial biopsy revealed ISHLT Grade 3R cellular rejection. She also developed strong donor specific antibody against DQ7. This episode of rejection was refractory to aggressive therapy including pulse steroids, Thymoglobulin (10 doses), Rituximab (4 doses) and IV immunoglobulin therapy. She received Alemtuzumab (Campath™-1H) 30 mg × 1 dose for steroid and thymoglobulin-resistant rejection. She received a second OHT due to severe cardiac allograft dysfunction two weeks after receiving Campath™.
In the outpatient setting following her second OHT, she experienced severe immunosuppression with delayed recovery of her CD4 lymphocyte count. She remained on anti-bacterial, anti-mycobacterial, anti-fungal, and anti-viral prophylaxis due to a persistent severely decreased cell count compared with age appropriate normal values (Figure 1) [2]. She presented to the outpatient solid organ transplant (SOT) clinic 10 months after her second OHT with a linear white oral lesion that extended in a horseshoe shape from the right lower to the left lower buccal surfaces, with intermittent extension onto the lips. At this time her CD4 count varied between 90 and 140 cells/µL. The lesion had developed insidiously, could not be easily scraped away with a tongue depressor (Figure 2a), was painless and did not cause any itching or irritation.

Figure 1: Absolute lymphocyte and CD4 cell count before and after Campath™ therapy.
She was initially managed with an empiric 10 day course of oral fluconazole therapy with no improvement, at which time the infectious disease team was consulted. We considered both common etiologies of white oral lesions in the setting of severe immunosuppression and lesions associated with depressed CD4 count more typical of that seen in patients with AIDS (Table 1), including hairy leukoplakia and Kaposi’s sarcoma. Fungal cultures and Herpes simplex (HSV) viral antigen detection by PCR were negative. We proceeded to a biopsy early in her evaluation based on her depleted CD4 count and vulnerability to the above mentioned lesions not typically seen in standard SOT immunosuppressive regimens. A biopsy performed by the otolaryngology team ultimately revealed parakeratosis with bacterial colonization and features suggesting chronic biting of the oral mucosa or Morsicato mucosae oris (Figure 2b). Staining for human papilloma virus (HPV) was negative. The lesions resolved after 8 weeks with the aid of a soft mouth guard and behavioral modification therapy.
Campath™ is a humanized rat monoclonal antibody directed against the CD52 antigen. The CD52 antigen is found on peripheral lymphocytes, thymocytes, natural killer cells, monocytes, macrophages and the lining of the male reproductive tract. There is no detectable effect on plasma cells, memory cells, granulocytes or platelets, erythrocytes or progenitor cells. It is highly potent for both T-cell and B-cell suppression resulting in more than 99% depletion of peripheral lymphocytes. The recovery of B lymphocytes occurs after 3-12 months and T-lymphocytes (CD4 and CD8) recover in a much more protracted manner, often requiring more than 2 years. The recovery of CD4 positive helper T-cells and absolute white cell count of this patient were significantly delayed (Figure 1). Pediatric experience with Campath™ therapy in OHT recipients has been limited, but is increasing [1].
The incidence of various oral lesions in adult SOT populations has been described in many studies, whereas the incidence in pediatric SOT populations has not been well described. The most common oral lesion seen in the SOT patient is drug-induced gingival overgrowth (DIGO). The incidence of DIGO is increased two fold in patients treated with cyclosporine compared with those treated with tacrolimus [3]. There are conflicting data concerning a causal relationship of gingivitis and poor oral hygiene to DIGO. Small case series have reported success in treating DIGO with transition from cyclosporine to tacrolimus [3]. Common infectious causes include Candida albicans and gingivostomatitis caused by herpes simplex virus (HSV). The incidence of oral candidiasis in a recent large survey of 500 renal transplant patients was 7.5% as compared with 4.2% of healthy controls [4]. Previous reports range from 9.4-46.7%, with a large portion of cases attributed to denture-associated candidiasis, a condition not applicable to the pediatric population [4]. Testing for HSV infection in mouth lesions is important in the SOT patients with reported incidence ranging from 0%-13%, median 2.6% as compared with 2.4% in healthy controls in the cited adult renal transplant population because treatment with acyclovir will be beneficial [4]. The findings on examination for our patient were inconsistent with HSV gingivostomatitis as her lesions were painless. Hairy leukoplakia, associated with Epstein Barr Virus, is a white corrugated, painless lesion most commonly seen on the lateral sides of the tongue and may occur in 4% of adult OHT patients and varied frequency in renal transplant patients of 1.5-11% [4]. Other oral lesions seen in renal transplant patients include apthous ulcers, lichen planus and xerostomia [4]. Adult SOT patients are at much higher risk for lip cancers than the general population with relative risks of 16.8 to 65.6, and be may be increased in pediatric SOT patients who are likely to experience both more time in the sun and a longer potential lifespan for exposure [4]. An oncologic process of concern in our patient was the possibility of HPV or HPV induced oral cancer based on prolonged and severely diminished immune surveillance capacity. Oral cancers in the adult SOT patient population occur with a relative risk of 2 to 4 as compared to the general population [2]. Since our patient experienced prolonged T-cell depression, Kaposi’s sarcoma was also considered. Kaposi’s has been reported in SOT patients in case reports and is caused by HHV-8 [3].
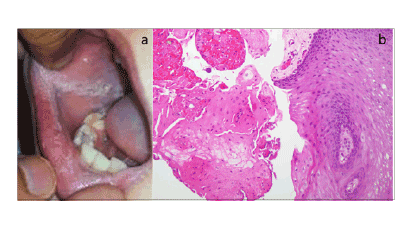
Figure 2a:: Oral lesion seen at the time of diagnosis. Lesion extends
onto the left lower buccal surface similar in appearance to the right.
Figure 2b: Histology (H &E staining of mucosa 20x) showing nucleated keratinocytes in the stratum corneum with no nuclear atypia and is suggestive of benign parakeratosis.
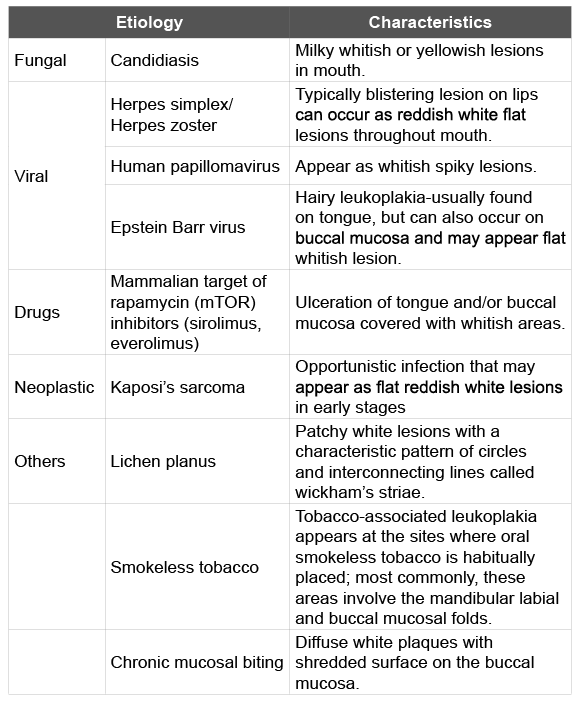
Table 1: Differential Diagnosis of white oral lesions associated with immunosuppression
Chronic biting of oral mucosa or Morsicato mucosae oris is an innocuous self-inflicted injury that is occasionally seen in children suffering from developmental or psychological problems [5]. Repeated biting leads to a chronically traumatized area, which is sometimes thickened, scarred and present as white lesions that may or may not be tender. A broad spectrum of oral conditions such as lichen planus and smokeless tobacco lesions may mimic the biting lesions. The management strategies vary from counseling to prescription of prosthetic shields [6]. In our patient, a soft mouth guard and counseling with behavioral therapy was successful in managing the hyperkeratosis associated with biting of oral mucosa and the lesions resolved after 2 months.
This case illustrates the finding that while SOT patients who are immunosuppressed are at risk for rare complications, they may also present with common conditions such as biting of oral mucosa. Before treating any oral mucosal lesion in the setting of immunosuppression, it is advisable to determine if there is an underlying cause that would respond to anti-infective treatment. Obtaining tissue for histopathology in order to rule out infectious causes is critical for developing an effective treatment plan. Interventions that will improve persistent biting lesions of the oral mucosa may also be considered for lesions that have the appearance consistent with Morsicato mucosae.
All authors contributed equally to this manuscript. Kevin Engelhardt and Bibhuti Das contributed to the concept and drafting the article; Jane Siegel contributed to critical review of the article.
Download Provisional PDF Here
Article Type: Case Report
Citation: Engelhardt K, Siegel J, Das BB (2015) A New White Oral Lesion in a Pediatric Heart Transplant Recipient Following Campath Therapy: Expanding the Differential Diagnosis. J HIV AIDS 1(2): http://dx.doi.org/10.16966/2380-5536.109
Copyright: © 2015 Engelhardt K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access