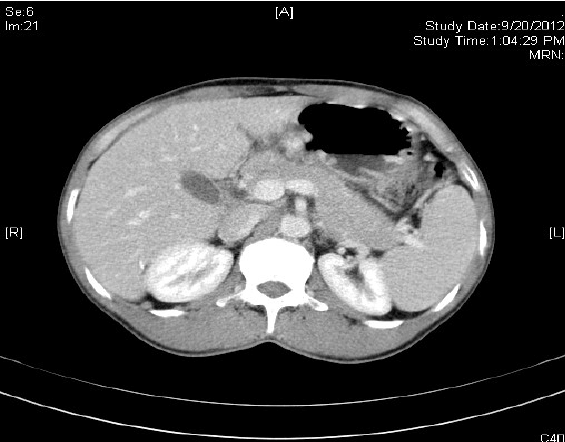
Figure 1: CT image showing irregular nodular thickening of the posterior wall of the body of the stomach along the lesser curvature.
Myla Yacob* Chandran B Sudhakar Samarasam I
1Department of Upper GI and General Surgery, Christian Medical College and Hospital, Vellore, Tamilnadu, India*Corresponding author: Myla Yacob, Department of Upper GI and General Surgery, Christian Medical College and Hospital, Vellore, Tamilnadu, India, 632004, E-mail: mylayacob@cmcvellore.ac.in
Introduction: Neuroendocrine tumors develop throughout gastrointestinal tract. The concurrence of mixed adenocarcinoma and neuroendocrine tumor together in the stomach is rare.
Case report: We report two cases of poorly differentiated carcinoma stomach with neuroendocrine component. These patients underwent radical total gastrectomy with D2 lymph node dissection followed by adjuvant chemotherapy. They are asymptomatic and disease free on follow -up.
Conclusion: Combination of neuroendocrine tumours and adenocarcinoma stomach a rare entity. Pre operative diagnosis with endoscopic biopsy is difficult. However, if there is a suspicion based on endoscopic biopsy, and then Immunohistochemical staining for neuroendocrine tumor markers may help in establishing preoperative diagnosis.
Stomach; Neuroendocrine cell carcinomas; Adenocarcinoma; Composite tumour
Neuroendocrine tumors of the gastrointestinal tract is rare, they account less than 1% of GI malignancies [1]. The combination of both adenocarcinoma and neuroendocrine tumor together in the gastrointestinal tract is very rare. Gastric neuroendocrine carcinoma arises predominantly from endocrine precursor cells that develop in the preceding adenocarcinoma component. Immunohistochemical staining of the tumor cells are positive for one of the neuroendocrine tumor markers such as chromogranin A, synaptophysin. Adenocarcinoma with neuroendocrine tumors have poor prognosis due to high proliferating biological behavior. Here we report a case of carcinoma stomach, histologically showed poorly differentiated adenocarcinoma with concurrence of neuroendocrine tumor.
A 57-year-old man presented with abdominal pain and melaena for six months. He had decreased appetite and significant weight loss. He had normal bowel habits. He was a reformed smoker. He was on regular medications for hypertension. Clinical examination was unremarkable. Blood investigations were normal. Oesophago gastroduodenoscopy showed an ulcer proliferative growth in the body along the lesser curve of the stomach, duodenum was normal (Figure 1). HPE of the biopsy was poorly differentiated adenocarcinoma. Contrast enhanced computed tomography of the abdomen and pelvis showed a large, exophytic lesion involving hepatoduodenal ligament, no metastasis to the liver. He underwent total gastrectomy with D2 lymph nodal dissection and Rouxen-Y anastamosis.
The histopathological examination of the surgical specimen showed poorly differentiated mixed adeno –neuroendocrine carcinoma infiltrating the muscularis propria (Figure 2). Two out of fifteen nodes had metastsic tumor. Tumor cells were synaptophysin positive, focal faint positive for chromogranin and CD 56 (Figure 3). MIB proliferative index was about 50-60%. He received six cycles adjuvant chemotherapy. He was asymptomatic and disease free on follow- up of two years.
A 42- year-old man was evaluated for upper abdominal pain and loss of appetite for duration of three months. He had had significant weight loss and a few episodes of non bilious vomiting. He had no known comorbidities. Clinical examination was normal except for visible gastric peristalsis. He was found to have ulcerative growth in the antrum and scope could not be negotiated into the duodenum (Figure 4). Histopathological examination revealed poorly differentiated adenocarcinoma. There were no liver metastases or ascites on CECT of the abdomen. A D2-sub-total gastrectomy was done with Roux-en-Y anastamosis.

Figure 1: CT image showing irregular nodular thickening of the posterior wall of the body of the stomach along the lesser curvature.
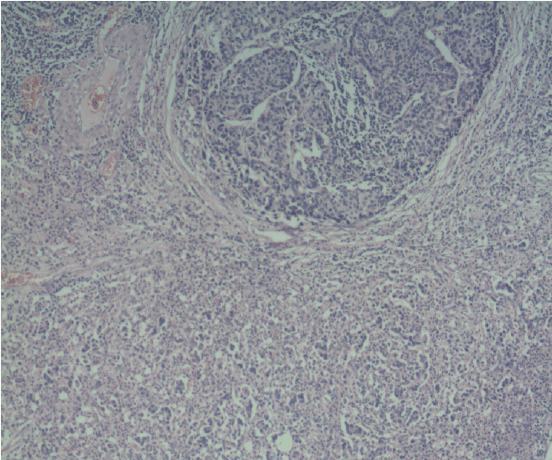
Figure 2: HE.100x (HE: Hematoxylin and eosin) shows mixed adenoneuroendocrine carcinoma component.
Surgical specimen’s HPE conclusive of poorly differentiated mixed adeno-neuroendocrine carcinoma infiltrating the serosa. The pathological staging was pT4N2Mx and tumor cells were positive for synaptophysin, chromogranin, and MIB proliferative index was 40%. He was given six cycles of adjuvant chemotherapy and was disease free on follow up for six months.
Neuroendocrine tumors can develop throughout the gastrointestinal tract, more common in the small bowel, rectum and stomach, rarely in the esophagus and anus. The combination of both adenocarcinoma and neuroendocrine tumor together in the gastrointestinal tract is rare. A few cases have been reported in the literature. Adenocarcinoma and neuroendocrine carcinomas are each well known to occur in the background of atrophic gastritis [2] (Figures 3 and 4).
Various nomenclatures have been used to describe the concurrence of both adenocarcinoma and neuroendocrine cell carcinomas. They include composite carcinoid, mucin-producing carcinoid, argentaffin cell adenocarcinoma, goblet cell carcinoid, adenocarcinoid, small cell undifferentiated carcinoma. The first description of a gastrointestinal tumor with an exocrine and a neuroendocrine component was published by Cordier in 1924 [3].
Since 1987, exocrine- neuroendocrine cell carcinomas were classified into different subtypes. Lewin classified into composite, amphicrine and collision tumors [4]. In the WHO classification of endocrine tumours, exocrine-neuroendocrine were defined as ‘mixed exocrine-endocrine’ tumors. In the 2010 revision of the WHO classification, five categories were named, based on cellular atypia, nuclear abnormalities, the number of mitotic figures and the Ki-67 labelling index. According to the revised classification, these neoplasms are called “mixed adenoneuroendocrine carcinomas” (MANECs) [5].
More recently, Fujiyoshi et al. [6] reclassified mixed endocrine and nonendocrine epithelial tumors by dividing the tumors into six categories: 1) neuroendocrine cells interspersed within carcinomas; 2) carcinoids (neuroendocrine tumors [NETs]) with interspersed non-endocrine cells; 3) composite glandular-neuroendocrine cell carcinomas containing areas of carcinoid and conventional carcinomas; 4) collision tumors in which NETs and conventional carcinomas are closely juxtaposed, but not admixed; 5) amphicrine tumors predominantly composed of cells exhibiting concurrent neuroendocrine and non-endocrine differentiation; and 6) combinations of the previous types.
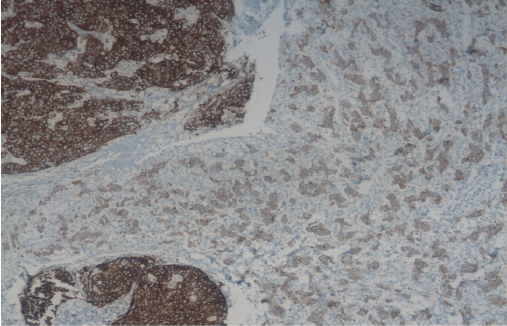
Figure 3: Immunohistochemical staining for synaptophysin highlights the neuroendocrine component of the tumour.
Figure 4: CT image showing circumferential asymmetric wall thickening is noted involving the pylorus of stomach and pyloroduodenal junction.
Most gastric endocrine cell tumors arise in the setting of hypergastrinemia causing the patient to present with peptic ulcer disease or chronic gastritis [2]. Tumours from the pylorus and antrum may present with symptoms of gastric out let obstruction.
The biological behaviour of mixed adeno neuroendocrine carcinoma of the stomach is significantly more aggressive than that of gastric adenocarcinoma [7]. Therefore preoperative diagnosis may help in assessing prognosis and planning appropriate treatment. These tumors are usually reported as poorly differentiated or undifferentiated tumors on endoscopic biopsy. Therefore appropriate immunohistochemistry may be performed in suspicious cases, in order to achieve a preoperative diagnosis [8].
The histological origin of these composite tumors is unclear. It has been postulated that signet-ring cell carcinomas originate from the gradual dedifferentiation from enterochromaffin-like (ECL) cells through signet ring cells with endocrine immunoreactivity [9]. There are two other hypotheses have been proposed, the first suggesting coincidental neoplastic changes in two different cell types and the second proposing a single common precursor cell that undergoes a bidirectional neoplastic change [10].
Composite neuroendocrine carcinomas with adenocarcinomas in the stomach can be diagnosed if at least one of the neuroendocrine markers, such as chromogranin A, synaptophysin, and NSE has a positive reaction in the immunohistochemical staining of the neuroendocrine carcinoma component [11]. In our case, the neuroendocrine carcinoma had a strong positive response to synaptophysin positive, focal faint positive for chromogranin and CD 56. MIB proliferative index was about 50-60%.
Treatment of composite tumors of adenocarcionoma with neuroendocrine cancers that occur in the stomach is radical surgical resection with D2 lymph node dissection and Roux-en-Y anastamosis including the tumours which are less than 2 cm. There is no role for endoscopic resection unlike smaller neuroendocrine tumours of the stomach. In patients with locally advanced adenocarcinoma with type 2 NETs of the stomach secondary to ZES/MEN I syndrome, neo-adjuvant chemotherapy including somatostatin analogs can be initiated for tumor regression [12]. In cases of solitary liver metastasis, radical gastrectomy with hepatectomy is effective treatment [13]. An early metastasis to lymph nodes and liver associated with neuroendocrine tumors due to their aggressive biological behavior. Intensive chemotherapeutic regimens, including cisplatin, doxorubicin, and vincristine can be administered. The adjuvant chemotherapy after gastrectomy has significantly reduced liver and lung metastasis [14]. In patients with multiple metastases confined to liver who are well preserved, and have a patent portal vein, Hepatic arterial embolization is recommended as a palliative option [15].
Because of uncertain histopathogenesis and rare occurrence, the clinical behavior and prognosis of composite tumors is still unclear, but Volante et al. [7] reported that the clinical behavior of composite carcinomas depends on the adenocarcinomatous component if the associated endocrine component is well-differentiated and upon the neuroendocrine component if it is poorly-differentiated. Adenocarcinoma with an endocrine component is more aggressive than ordinary adenocarcinoma [8].
Combination of neuroendocrine tumours and adenocarcinoma stomach not a rare entity. Pre operative diagnosis with endoscopic biopsy is difficulty due to focal lesions. Immunohistochemical staining is advised in poorly differentiated adenocarcinoma of the stomach. Neuroendocrine tumors associated with poor prognosis due to high proliferative behaviour.
Download Provisional PDF Here
Aritcle Type: Case Report
Citation: Yacob M, Sudhakar CB, Samarasam I (2015) Composite Glandular and Neuroendocrine Carcinoma of the Stomach- Two Case Reports. J Gastric Disord Ther 1 (2): http://dx.doi.org/10.16966/2381-8689.110
Copyright:© 2015 Yacob M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access