Abstract
Introduction: Bisphosphonates are widely used in the treatment of bone disease due to their inhibitory effects on bone remodeling. Although it is well established that bisphosphonates act by direct effects on osteoclastic cells, there has been increasing evidence suggesting that they may also work on osteoblast cells. The reported effects of these drugs on osteoblast cells are conflicting with increasing number of studies suggesting that at different concentrations, and with different types of bisphosphonates osteoblast differentiation and bone formation activities are varied. Side effects such as osteonecrosis of the jaw are seen with chronic use of bisphosphonates. To better develop approaches to minimize these
adverse effects it is important tofurther understand the effects of bisphosphonates on osteoblasts and their modulation by endogenous regulatory
factors.
Materials and Methods: Human alveolar osteoblastic cell cultures were treated with the bisphosphonate, alendronate, platelet derived growth
factor and a combined treatment of alendronate and platelet derived growth factor. Cell activity was assessed with a mitochrondrial enzyme
assay, and differentiation with spectrophotometric assays for alkaline phosphatase and mineralization over a period from 24 hours to 17 days.
Results and Conclusion: Treatment of the osteoblastic cells with alendronate (10-8 M) produced small, significant effects on cell activity and
markers of differentiation that varied with the time of incubation. The effects of platelet derived growth factor on these same parameters were
maintained with co-incubation with alendronate suggesting this growth factor may have a therapeutic role in the minimization of the negative
side effects of the drug. These data are supportive of the emerging potential of the clinical use of platelet growth factor enriched plasma for
bisphosphonate-induced osteonecrosis of the jaw.
Keywords
Alendronate; Bisphosphonates; Osteoblasts; Platelet-derived growth factor; Osteonecrosis
Abbreviations:
BP: Bisphosphonate; ALD: Alendronate; PDGF:
Platelet-derived growth factor; BRONJ: Bisphosphonate related
osteonecrosis of the jaw
Introduction
Bisphosphonates (BPs) are drugs widely used in the treatment of bone
diseases such as osteoporosis, Paget’s disease, hypercalcemia associated
with malignancy, bone metastasis and loss accompanying multiple
myeloma and inflammatory conditions [1-6].
It is well established that BPshave inhibitory effects on bone remodeling
via direct effects on osteoclasts, the main bone resorptive cell type [7].
However, since there is much evidence that osteoblasts, the main bone
forming cell type, are also involved in the regulation of osteoclastogenesis,
the effects of BPs may not be solely onosteoclastic cells. Osteoblasts are
involved in osteoclast regulation via their production of nuclear factor
(NF)-ĸB ligand (RANKL) [8]. Osteoblasts have also been widely shown
to produce osteoprotegerin (OPG), a soluble receptor for RANKL, that
acts as a decoy to competitively inhibit the binding of RANKL to RANK
and inhibits RANK activation and thus decreases osteoclast activation
[9]. Consistent with these studies are those that have shown that BPs can
inhibit the expression of RANKL and increase the expression of OPG in
osteoblastic cells [10,11].
Although these results implicate the involvement of osteoblastic cells
in the antiresorptive action of BPs via RANKL signaling [10,11], other
effects of these agents on osteoblastic cells have not been consistently
observed as presented in a recent review [12]. It has been suggested that
the conflicting results in osteoblastic cell studies might be due to the
bisphosphonate being studied, its effective concentration, as well as the
osteoblastic cell type [12]. For example, positive effects of BPs on markers
of osteoblastogenesis have been noted with concentrations from 10-9 to 10-
6
M but inhibitory effects at higher concentrations [13-22]. A recent study
with alendronate (ALD) shows that this BP affects osteoblasts indirectly
through the ephrinB1-EphB pathways involved in the cross talk between
osteoclasts and osteoblasts. This study with rat bone marrow cells provides
in vitro evidence that ALD acts directly on osteoclastic cell precursors,
which then act on osteoblastic cell precursors to inhibit osteoblastic
differentiation and related bone formative activities [23].
Although chronic use of BPs has been associated with side effects such
as atypical femoral fractures and osteonecrosis of the jaw, these drugs are
still widely used because of their generally positive therapeutic effects on
management of fractures and associated bone pain.ALD is often the firstline
therapy choice for prevention of osteoporotic fractures but it might
impart a higher risk of atypical femoral fractures and osteonecrosis of the
jaw than other orally administered BPs [24]. In order to develop strategies
to minimize the negative effects of BPs it is imperative to have a better
understanding of the mechanism of action of these drugs. Toward this end,
the effects of ALD on primary cultures of osteoblastic cells from alveolar
bone specimens were studied here with a focus on modifications of the
responses in the presence of platelet derived growth factor, (PDGF). The
therapeutic implications of these responses in BP induced osteonecrosis
of the jaw (BRONJ) are discussed.
Materials and Methods
Alendronate (ALD) was purchased as alendronate sodium from Sigma
-Aldrich (St Louis, MO). The platelet-derived growth factor (PDGF) used
here was human recombinant PDGF-BB. This and all other reagents were
also purchased from Sigma-Aldrich unless otherwise stated.
Human osteoblastic cells were obtained from alveolar bone specimens
using conditions previously described [25]. The use of these specimens,
that would have been otherwise discarded, was according to guidelines
of the University at Buffalo’s Human Subjects Institutional Review Board.
Mineralization assay
The osteoblastic cells were seeded at a concentration of 30,000 cells
per well, in a 24 well tissue culture plate. The cells were cultured with
ALD (10-8 M), PDGF (10-8 M) or a combination of the two agents with
controls having no added agent in alpha-minimum essential medium
(MEM, Gibco, Life Technologies, Grand Island, NY) supplemented
with heat activated 10% fetal calf serum, 1% L-glutamine, penicillin G,
streptomycin sulfate, and amphotericin B at 37°C with 5% CO2
. In most
experiments, the cultures were incubated with replacement of media and
treatment solutions every two days. Total incubation periods varied from
10-21 days.
Mineralization was quantified with a colorimetric Alizarin red assay
that measures the calcium mineral content associated with the cell
cultures using an adaptation of the protocol of Gregory et al [26]. Cell
monolayers were washed with phosphate-buffered saline (PBS) and fixed
with cold 70% ethanol for one hour. The monolayers were rinsed with
highly purified water before the addition of 40mM Alizarin red S (ARS), at
pH 4.2. After all the dye was absorbed into the monolayers the wells were
each washed 5 times with purified water and once with PBS. The plates
were stored at -200
prior to dye extraction. Cetylpyridinium chloride
(CPC) extraction was used for the destaining. ARS was removed from the
monolayers by the addition of CPC (10% w/v, pH 7.0). The plates then
were incubated at room temperature with gentle shaking for 1 hour. The
absorbance of the CPC extractions was measured at 550 nm [26].
Alkaline phosphatase activity
The human osteoblastic cells were incubated with ALD and PDGFas
described above for time periods from 24-72 hours, and analyzed for
ALP activity as an indicator of osteoblastic cell differentiation using the
para-nitrophenol phosphate assay previously described in detail with
normalization of the data on the basis of associated total cell protein [27].
Tetrazolium salt (MTT) assay: This assay was used to assess
osteoblastic cell activity. The primary human osteoblastic cells were
incubated with ALD, PDGF or combinations of these agents as described
above for various time periods. At the end of the experimental period (24,
48 or 72 hrs), MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium
bromide reagent was added to the cells for 4 hrs and the assay conducted
as previously described in detail [28].
Results and Discussion
Figure 1 shows that the effects of ALD (10-8 M) on osteoblastic cell
activity varied over the course of time of incubation with small, but
significant decreases compared to controls after 24 hours of incubation,
no effects after 48 hours and small, but significant increases after 72 hours.
At all of these time periods, PDGF (10-8 M), produced increases in activity
compared to controls and these PDGF-induced increases were not altered
in cells cultured in combination with ALD (10-8 M).
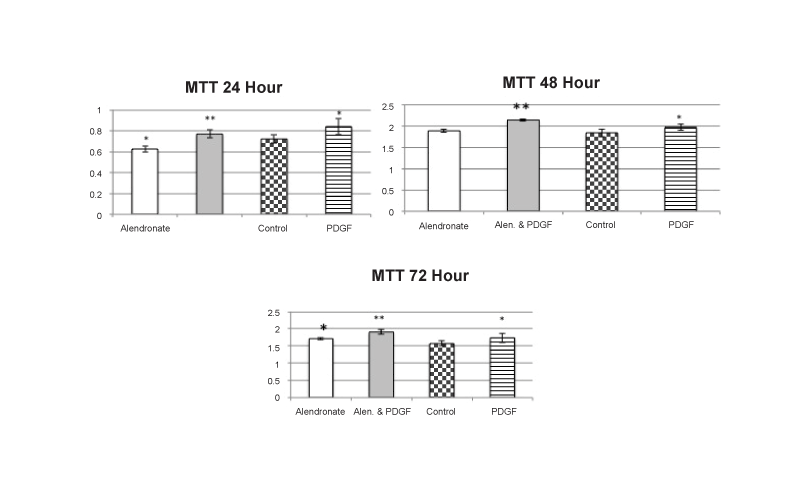
Figure 1: MTT Cell Activity
After 24 hours of incubation, alendronate produced small, significant decreases in cell activity compared to controls and coincubation of alendronate
and PDGF resulted in levels significantly greater than with alendronate alone and not significantly different than PDGF alone. After 48 hours there
was no effect of alendronate alone compared to controls with PDGF and alendronate together still exhibiting the PDGF induced increases. Small,
but significant increases were observed with alendronate after 72 hours with the PDGF induced increase not significantly altered by coincubation
with alendronate. Values are the mean +/- SEM with n=4 samples per group: * = significantly different from control; ** = significantly different from
alendronate alone; = p<0.05 ANOVA.
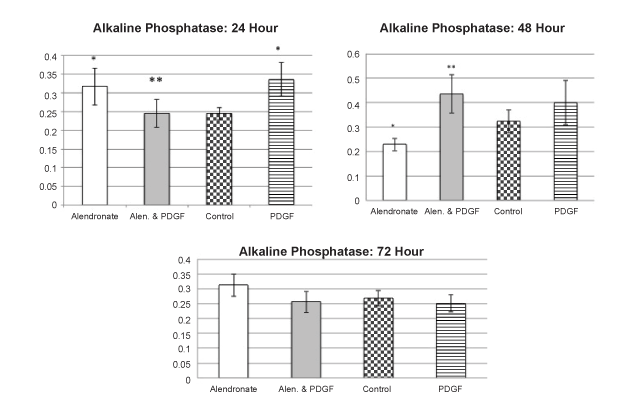
Figure 2:Alkaline Phosphatase Studies
After 24 hours, alendronate produced significant increases in alkaline phosphatase activity compared to controls. PDGF alone produced similar effects
but in combination with alendronate there was a significant reduction in this effect to control values. After 48 hours, alendronate significantly decreased
alkaline phosphatase levels compared to all groups. The combination of PDGF and alendronate significantly increased alkaline phosphatase compared
to alendronate alone. After 72 hours. No significant effects of either alendronate or PDGF were observed. Values are the mean +/- SEM with n=4
samples per group: * = significantly different from control; ** = significantly different from alendronate alone; = p<0.05 ANOVA
The effects of ALD and PDGF on ALP activity were also time
dependent. Figure 2 shows that after 24 hours of incubation with ALD
(10-8 M) there was a small, but significant increase in this early marker
of osteoblastic cell differentiation. PDGF (10-8 M) produced a similar
increase over controls. However, at this time period, incubation with the
combination of both of these agents, each at 10-8 M, resulted in no effect
on ALP activity compared to controls. After 48 of incubation with ALD
there was a significant decrease in ALP compared to controls. Although
the effects of PDGF alone at 10-8 M or in combination with ALP were not
significant increases compared to controls, they were significantly greater
than ALD alone. After 72 hours, there were no significant effects on ALP
activitywith any treatment group compared to controls.
Figure 3 shows that a 10-day incubation with ALD (10-8M) resulted
in significant decreases in mineralization in the human osteoblastic cell
cultures compared to controls. During this same time period, PDGF
(10-8M)-treated cells had significant increases in mineralization. When
cells were incubated with both agents each at a concentration of 10-8 M for
the 10-day period, the PDGF-induced increases in mineralization were
not significantly altered by the ALD treatment. ALD-induced decreases
in osteoblastic cell mineralization were noted when incubations were
conducted up to 17 days (data not shown). The results shown in Figure
4 suggest that ALD may not need to be present for the entire duration
to achieve a significant decrease in mineralization in this cell system.
Incubation with ALD (10-8M) for 7 days, followed by removal of the drug
containing media and replacement with fresh media and incubation for
an additional 5 days resulted in a similar decrease compared to controls
as when the cells were incubated with ALD for the entire 12-day period.
When PDGF (10-8M) was added to cultures in which the ALD was
removed, there was a significant increase in mineralization during the
subsequent 7-day incubation period.
Although there are several BPs presently used for therapeutic
management of skeleton related conditions, in this present study the
effects of ALD on human alveolar osteoblastic cells was the focus for
several reasons. ALD is often the main line choice for oral antiosteoporotic
therapy [24]and there are data that suggest that adverse skeletal side
effects such as osteonecrosis of the jaw and atypical femoral fractures may
be higher with ALD compared to other orally administered BPs such as
risedronate, ibandronate, etidronate and clodronate [24].
The underlying mechanisms involved in the adverse side effects on
bone are not understood, but it has been reported that ALD has a greater
affinity for the tissue along with more significantdecreased bone turnover
as well as greater anti-angiogeniceffects in comparison with other BPs that
are given orally [29,30].
The results reported here are consistent with the growing body of
studiesthat suggest that ALD can have direct effects on osteoblastic cells
and that these effects can vary with the time of incubation and various
associated factors [12]. The studies presented here focused on the possible
interaction between ALD and PDGF primarily because there is some
evidence that PDGF may have some therapeutic value in the healing
process in BP- induced osteonecrosis of the jaw [31].
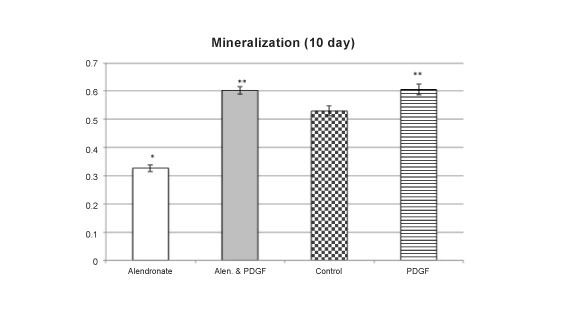
Figure 3: Mineralization Studies
After 10 days of incubation, alendronate produced significant decreases
in mineralization compared to controls. Incubation with PDGF for this
same period produced significant increases that were not altered with
coincubation with alendronate. Values are the mean +/- SEM with n= 4
samples per group: * = decreases compared to controls; **= increases
compared to controls as well as alendronate alone; p<0.05 ANOVA
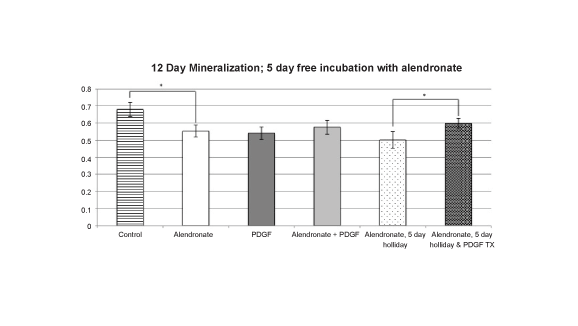
Figure 4: Mineralization Studies with an Alendronate free period
After 12 days of incubation, the alendronate group showed a significant
decrease in mineralization. A five-day drug free period (holiday) from
the alendronate had no significant effect on mineralization compared to
alendronate present for the entire period. The treatment of PDGF during
the alendronate free (holiday) period increased mineralization significantly
compared to the alendronate 5 day free treated group without the added
PDGF. Values are the mean +/- SEM with n= 4 samples per group: * =
significant differences; p<0.05 ANOVA
PDGF is present in bone matrix, synthesized not only by platelets, but
also monocytes, macrophages, endothelial cells and osteoblasts [32]. The
growth factor molecule is a dimer that can be made from four difference
polypeptide chains (A,B, C, D). From the different possible combinations
of these chains, PDGF-BB appears to be the most biologically potent in
the skeleton and has been shown to bind to osteoblasts with the highest
affinity [33, 34].It has been shown to be produced at fracture sites and
to be present during the early stages of fracture repair [35]. In a rat
model, systemic administration of PDGF not only prevented the loss of
bone normally induced by ovariectomy, it also maintained bone strength
throughout the skeleton. Co administration of PDGF and alendronate in
this animal model resulted in bone density levels greater than that seen
with either agent alone. These data suggest that PDGF may be effective
in producing anabolic effects on bone even in the presence of the
bisphosphonate and potentially inhibited bone remodeling activity [36].
Studies have shown that PDGF-BB stimulates chemotaxis and
proliferation in osteoblasts and increases collagen synthesis by this cell
type [33]. The direct effects of PDGF on differentiation parameters such
as alkaline phosphatase and mineralization appear to be more variable
depending upon exposure conditions. A study focusing on the expression
of these parameters in vitro showed thatshorter-term exposure to PDGF
produces increases where asin longer-term incubation there are decreases
in these parameters [34]. Based on these observations, it appears that
increases in bone formation seen in several in vivo studies are largely
due to the increased proliferative effects on osteoblastic cells [34].The
studies presented here support the temporal effect of PDGF on alkaline
phosphatasewith increases at the earliest measured time period of 24
hours and decreases or no significant effects after longer periods of 48 or
72 hrs. Likewise, PDGF’s effects on mineralization were increases after
10 days of incubation, but after 12 days, decreases were observed. The
stimulatory effects of PDGF on cell activity observed here are consistent
with increases in proliferation over sustained periods of time. Of
particular interest to the potential use of PDGF as a therapeutic agent to
restore bone healing in BRONJ is that the combination of ALD and PDGF
in the present study restores the ALP induced decrease in osteoblastic cell
activity after 24 and 48 hours of incubation. In the mineralization study
it is particularly interesting to note that after ALD is removed from the
osteoblastic cell cultures for 5 days after a 7-day incubation, the addition
of PDGF restores the decreased mineralization marker levels to control
levels at the end of the total 12-dayperiod. These results are consistent with
a previous report that osteoblastic cells isolated from patients with BPinduced
osteonecrosis responded to PDGF in a positive manner similar
to cells isolated from alveolar bone of persons not treated with BP [37].
As recently reviewed [38] there have been a number of reports on
the therapeutic effects of PDGF on the regeneration of alveolar bone,
periodontal tissues as well as wound healing in general[39-43]. Local
applications of PDGF-BB have been shown to destabilize blood vesselsand
result in growth of new vasculature at the site of the healing wound [38].
Since PDGF has been documented to possess amultitude of effects that
promote bone and periodontal tissuerepair and regeneration it should
be a natural candidate for therapy inoral necrotic conditions although
it does not appear that it has been tested directly in this regard. There
have, however, been several reports of successful use of platelet rich
plasma (PRP) containing relatively high levels of PDGF in addition to
other growth factors for the treatment of BRONJ. Adornato et al. [44]
treated 12 patients with refractory BRONJ with a combination of bone
resection and autologous platelet-derived growth actors.After six months,
10 of the patients had complete recovery of mucosal and bony defects and
the remaining 2 showed some improvement in healing. Subsequently,
Mozzati et al. [45] reported successful treatment of 32 cases of BRONJ
(Marx IIB classification [46]) by application of PRP over the bony defect
after resection of the necrotic tissue. An update paper from this group
documented freedom from complications and need of reintervention
to be 100% in these patients after a 7-year follow up [47]. In addition,
another report of 32 successful cases of treatment of BRONJ with PRP is
found in the clinical review of Long et al. [48].
It is recognized that clinical use of platelet rich plasma (PRGF) can
offer advantages over the use of PDGF alone. PRP contains many growth
factors released from activated platelets in addition to PDGF such as
transforming growth factor-beta, endothelial growth factor, vascular
endothelial growth factor, insulin-like growth factor-1, basic fibroblast
growth factor and hepatocyte growth factor [49].
Studies have reported that bisphosphonates such as pamidronate
and zolendronic acid, given to cancerpatients before chemotherapy,
can produce significantdecreases in PDGF as well as angiogenic factors
such as vascular endothelial growth factor (VEGF) [50,51]. Decreases
of this nature in the concentrationsof factors that have significant effects
on osteoblastic andosteoclastic cells can influence the overall effects of
thebisphosphonates on bone remodeling and lead to anosteonecrotic
condition. Local application of PRP on BP induced osteonecrotic wounds,
may therefore have significant positive effects of healing of bone and
surrounding tissues via increased concentrations of PDGF as well as
angiogenic factors such as a VEGF at the compromised site as suggested
by the case reports of successful management of this condition reported
by an growing number of clinical investigators [44-49,52,53]. Although
the number of such cases reported in the literature has rapidly increased,
case- control randomized studies to support the use of PRP therapy for
BRONJ are still lacking [48].
Conclusion
Direct effects of alendronate on human alveolar osteoblastic cells
activity, ALP and mineralization were observed with both increases and
decreases depending upon the incubation conditions. hrPDGFmodulated
these effects in a manner consistent with what has been observed in
clinical reports on therapeutic effects of platelet rich plasma in BRONJ.
Article Information
Article Type: Research Article
Citation: Barres L, D.S. Mota Anna, Greenberg M,
Almojaly S, Dziak R (2015) Effects of Alendronate on
Human Alveolar Osteoblastic Cells: Interactions with
Platelet-Derived Growth Factor. Int J Dent Oral health,
Volume1.2: http://dx.doi.org/10.16966/2378-7090.108
Copyright:© 2015 Barres L et al. This is an
open-access article distributed under the terms
of the Creative Commons Attribution License,
which permits unrestricted use, distribution, and
reproduction in any medium, provided the original
author and source are credited
Publication history:
Received date: 14 April, 2015
Accepted date: 27
April, 2015
Published date: 05 May, 2015





