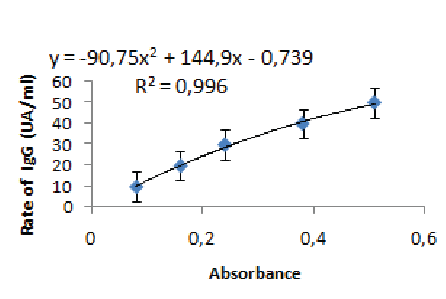
Figure 1: The equation of the trend line obtained from the range with MSP-2

Sourabié Y1,2* Bazié WW1 Sangaré I1,2 Sanou G5 Fumoux F4 Traoré Y3
1Centre Hospitalier Universitaire Sourô Sanou 01 BP 676, Bobo, 01 Burkina Faso*Corresponding author: Yacouba Sourabié, Enseignant- chercheur en Immunologie à Institut supérieur des sciences de la santé, Université polytechnique de Bobo Dioulasso, Centre Hospitalo-universitaire Souro Sanou, Service d’Immunologie et d’Hématologie, BP 676, Burkina Faso, Tel: 0022670710325; E-mail: yacourabie@yahoo.fr
Background: The purpose of our study is to establish the relationship between the rate of total immunoglobulin G in the sera of subjects with hemoglobin E or A genotype and resistance to infection by Plasmodium falciparum.
Methods: This is a prospective study over a period of five months on the sera of Vietnamese.
The serums were from whole blood collected in 2002 by venipuncture in subjects of study. The collected blood of each patient was centrifuged and the obtained serum was aliquoted into cryotubes and stored at 20°C for immunological studies (IgG assay). To maximize the sensitivity of ELISA test, we have coupled the antigenic peptides using glutaraldehyd with, highly polymerized poly-L-lysine as a resin. The enzymatic reaction then takes place in the dark for 30 min after addition of substrate before reading the results using a spectrophotometer (Opsys MR Dynex® Technology) set at the wave length of 405 nm.
Results: A total of 182 blood samples were taken from people belonging to 35 families. None of the subjects had clinical symptoms of malaria at the time of sampling. The distribution of the age is 3-79 years; the sex ratio (M/F) is 1.6. The proportions of hemoglobin are 42% of hemoglobin AA carriers and 58% of hemoglobin EA and EA carriers. In our different age groups, specific total IgG against antigenic peptides (MSP-1, MSP- 2 and SR-11.1) of P. falciparum show an age-dependent distribution. In subjects 3-19 years, the titer of specific total IgG against Plasmodium falciparum is lower than that of more than 20 years. Comparison of specific IgG against the antigenic peptides MSP-1, MSP-2 and SR-11.1 of P. falciparum in the age groups, gives a significantly higher average for MSP-1 compared to SR-11.1 and MSP-2 (Khi2=4.12) The rate of total IgG against the antigenic peptides at subjects with hemoglobin E carrier is higher than those of hemoglobin A (p=0.004).
Conclusion: We have shown that the response of specific total Ig G against the plasmodium antigens (MSP-1, MSP-2 and SR-11.1) is improved in patients over 20 years with hemoglobin E genotype compared to those with the AA genotype. In subjects under 20 years, no significant differences were obtained between the different groups of hemoglobin. MSP-2 and MSP-1 appear much more immunogenic than the SR11.1 in different age groups.
Hemoglobin E; MSP1; MSP2; SR11.1; ELISA; IgG
In West Africa, it has been shown that after long exposure to intense transmission; it gradually develops a non-sterilizing immunity against Plasmodium falciparum. Moreover, in these endemic areas, some individuals appear naturally protected against clinical malaria and/or severe forms of malaria [1]. Suggesting a genetic control of resistance to malaria clinical infection, it has been clearly demonstrated in murine models that genetic factors control Plasmodium infection [2,3]. Studies have clearly demonstrated the role of abnormal hemoglobin in resistance to malaria attacks. In this resistance sickle cell trait (Hb AS) due to a change of the beta chain of glycin (6 beta: Glutamic acid → Valin) is the most studied genetic abnormalities of RBCs [4-6]. Thus, it was shown that susceptibility to Plasmodium falciparum infection and malaria attacks are less frequent in carriers of Hb S or C [7-9]. However, the mechanisms mediating this resistance remain unclear.
It was suggested that the protective effect in hemoglobinopathies may be partly due to the inhibition of Plasmodium falciparum increase in the S parasitized erythrocytes [10,11] and might thus induce modulation of the immunoglobulin G response in carrier individuals of these hemoglobinopathies [4].
A recent proposal of protection against Plasmodium falciparum mechanism in the carrying hemoglobin C and S shows that it is linked to an antigen exposure PfEMP1 anomaly on the surface of infected sickle cell C or S [6]. Other immuno-epidemiological studies have also shown that the protection against malaria is directed at least partially by the antibody [12], with a high titer of Ig G directed against malarial antigens in situations Hb AS/SS/SC versus AA [9,13,14]. Thus, [15,16] showed that the cytophilic antibodies (IgG1 and IgG3) protect against Plasmodium falciparum by activating effecters cells through FcgRIIA while IgG4 and IgG2 block the protection mechanisms. And porting HbS, IgG3 response against MSP-2 may influence the decrease of the title of IgG2.
Another hemoglobin E (homozygous or heterozygous) due to a substitution of Glutamic acid for lysine at the 26th amino acid segment of A beta globin chain is frequent in South East Asia and has been described as conferring resistance to multiplication in the erythrocytes of Plasmodium falciparum and vivax [10,17]. Chotivanich et al. [18] showed that patients had low parasite density in the carrying hemoglobin E which allowed protection against severe manifestations of malaria. Very few studies have been conducted to show the involvement of hemoglobin E in modulating the immune response against variant surface antigens of different stages of Plasmodium falciparum or vivax.
Data exist and have shown the role of hemoglobin E in the resistance of patients in severe malaria cases and simple. The purpose of our study is to establish the relationship between the title of total immunoglobulin G in the serum of subjects with hemoglobin E or A genotype and resistance to infection by Plasmodium falciparum.
This is a prospective study over a period of five months on the serum of Vietnamese living in the commune of XaTanh in Quang Tri’s province and aged 3-79 years. The study took place at the Faculty of Pharmacy of the Timone, Marseille: Mixed Research Unit-MD3 (host-parasite relationship and immunogenetics Pharmacotherapeutic).
Our serums were from whole blood collected in 2002 by venipuncture in subjects of study. The collected blood of each patient was centrifuged and the obtained serum was aliquoted into cryotubes and stored at 20°C for immunological studies (IgG assay). The choice about the town Xa Thanh is justified by the fact that in this area there is a high prevalence of malaria and haemoglobinopathy E is high in this area.
Direct sensitization by synthetic peptides on the ELISA plate is generally inefficient and highly dependent on their size and charge. To maximize the sensitivity of our test, we have coupled the antigenic peptides using glutaraldehyde using, highly polymerized poly-L-lysine as a resin.
Principle: poly-L-lysine is coated on the plate that serves as an anchor by exposing the NH3+ group in its side chains with reactive functions CHO glutaraldehyd. The covalent bond formed after chemical reaction is then used to fix the coupling agent to the plate. A second reaction between the second aldehyde function of glutaraldehyd and a primary amino group (NH3+terminal or lateral) allows covalent attachment of plasmodial antigens (MSP-1, MSP-2 and SR-11.1) to the poly-L-lysine and thus better attachment to the ELISA plate. The amino function of glycine was then used to block the aldehyde functions. We used the following protocol:
Poly-L-Lysine is coated on the plate overnight at 4°C (40 µg/ml in carbonate buffer pH=9.6), 100 µl/well. The plate is washed three times with PBS 1X and then 100 µl/well of 1% glutaraldehyd diluted in PBS 1X buffer were incubated for 30 minutes at room temperature. The plate is then washed three times and the antigenic peptide solution (10 µg/ml diluted in PBS 1X buffer) was added to the wells (100 µl/well). The plate is incubated overnight at room temperature, washed three times and then the free reactive functional groups are blocked by 1 M glycine (in PBS 1X, 200 µl/well) for 1 h at room temperature. Finally, the plated was washed and saturated with PBS-3% Milk (2 h at room temperature, 250 µl/well). Then we followed the steps of:
Each stage after the saturation of the plate is preceded by three washes in PBS 0.05% Tween, 200 µl per well.
Samples containing specific antibodies to be tested are diluted 1/20 in PBS 1X, for the measurement of specific total IgG.
For each plate, standards were included by using the plasma pool of African subjects as the positive controls. Negative controls are established with self plasmas unexposed Europeans to Plasmodium falciparum and a “white” in which the plasma is replaced with PBS 1X dilution buffer. The standards were made by dilution in a range of ½.
The diluted plasma (1:20) are deposited at 100 µl/well and incubated for 2 h at room temperature.
Then the plate is coated with the goat‘s anti-human immunoglobulin G, a secondary antibody coupled to alkaline phosphatase (diluted 1/3000 in PBS 1X according to the manufacturer’s recommendations (Beckman Coulter®) at 100 µl / well and ‘incubated for 2 h at room temperature, stirring.
The development is carried out by depositing the substrate of the enzyme alkaline phosphatase, p-nitrophenyl phosphate (pNPP), prepared extemporaneously at the rate of 200 µl per well. The enzymatic reaction then takes place in the dark for 30 min after addition of substrate before reading the results using a spectrophotometer (OpsysMRDynex® Technology) set at the wave length of 405 nm.
The processing and analysis of data is done using the Excel 2007 software for tables and graphs.
At all absorbance values, it was subtracted from the absorbance of “white”.
To compare the values obtained between the various processes, we have standardized the values of the positive pool. This correction was then applied to the optical density of each serum.
The sample titration is carried out using a range obtained from pool of sera of Africans living in endemic areas. The following dilutions of factors were used: 20; 50; 100; 200 and 400. The measured absorbance for the 1/400 dilution was associated with the value of 10 AU/ml. The equation of the trend line obtained from the range allowed us to calculate for each absorbance measured its equivalent in AU/ml (Figures 1-3).

Figure 1: The equation of the trend line obtained from the range with MSP-2
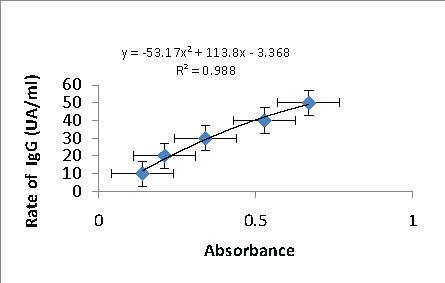
Figure 2: The equation of the trend line obtained from the range with SR-11.1
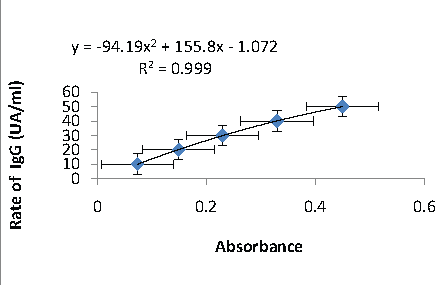
Figure 3: The equation of the trend line obtained from the range with MSP-1
The tests used for the comparison of means and proportions were the tests of chi-squared and Fisher, Kruskal Wallis. P values <0.05 were statistically significant.
Blood samples were taken in the town of Xa Thanh, Huong Hoa District. This town is located in a region above sea level, along the Sêpon river forms the border between Laos and Vietnam. The commune’s territory covers 1200 ha and includes 9 hamlets. The population at the last census has 2203 inhabitants and 382 families. All the inhabitants belong to ethnic Bru-Vân Kiêu. They are mainly farmers and herders. The families have on average 4.8 children, educational attainment remains low [19]. The Bru Van Kiêu practice intermittent crops, annually using burning zones remote from the permanent housing. They often spend the night in the forests. The habitat is mostly traditional; with the kitchen in the middle of the house (net use is limited due to fire fears). The people have very close relations with Laos and make frequent visits to Laos.
Huong Hoa district is area highly endemic malaria. More than 30 species of Anopheles (a total of 31 species in the province) have been described. The two main vectors of transmission are An. Minimus and An. Dirus. The majority of the catches of the province were carried out in the district of Huong Hoa (88% of An. Minimus and 95% of An. Dirus). The number of malaria cases is particularly strong during the rainy season (June to September). The data collected at the dispensary of Xa Thanh commune from 1997 to 2001 indicate an average of 125 cases of malaria per year for a population of 2203 people, with annual changes ranging from 72 to 194. Sixty percent of malaria cases are observed for four months, from June to September. There are P. falciparum infections (98%) and by P. vivax (2%). A total of 182 blood samples were taken from people belonging to 35 families. None of the subjects had clinical symptoms of malaria at the time of sampling. The distribution of the age is 3-79 years; the sex ratio (M/F) is 1.6.
We left people in four age groups: children 0-10 years, teens 11-19 years, young adults aged 20-31 years and adults walls more than 32 years. For each age group, we have a similar proportion of individual AA and AE. The phenotypes of hemoglobin of these subjects were determined by electrophoresis. The proportions of hemoglobin are 42% of hemoglobin AA carriers and 58% of hemoglobin EA and EA carriers.
In our different age groups, specific total IgG against antigenic peptides (MSP-1, MSP-2 and SR-11.1) of P. falciparum show an agedependent distribution. In subjects 3-19 years, the titer of specific total IgG against Plasmodium falciparum is lower than that of more than 20 years. Comparison of specific IgG against the antigenic peptides MSP-1, MSP-2 and SR-11.1 of P. falciparum in the age groups, gives a significantly higher average for MSP-1 compared to SR-11.1 and MSP-2 (chi2=4.12) (Figure 4).
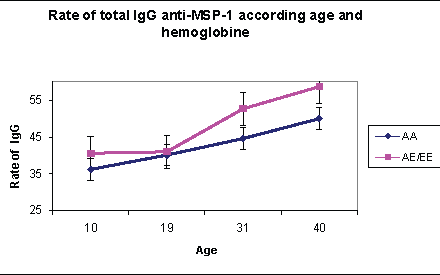
Figure 4: The effect of age and hemoglobin on the rate of anti-MSP- 1total IgG
The under 20 years are composed of 106 individuals whose average age is 11.7 years. The distribution of hemoglobin is 41/106 (39%) of hemoglobin AA carriers, (47/106) 44.3% of hemoglobin AE carriers and (16/106) 15.1% of hemoglobin EE carriers. In malaria’s endemic areas, acquired immunity is required in subjects in their contacts with the antigen and is dependent on the level of transmission. We observed that the rate of specific total IgG against P falciparum antigen such as MSP-1; MSP-2 and SR-11.1 obtained in the various groups of hemoglobin are not significantly different (p=0.07) (Figures 4-6).
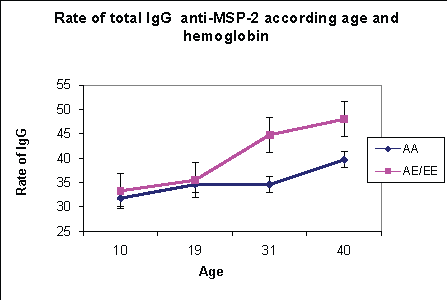
Figure 5: The effect of age and hemoglobin on the rate of anti-MSP- 2total IgG
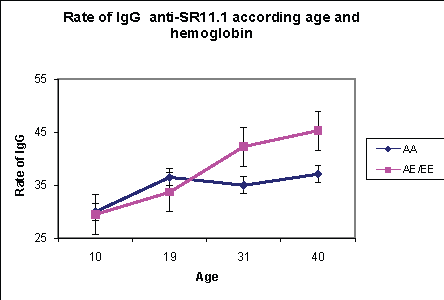
Figure 6: The effect of age and hemoglobin on the rate of anti-SR-11.1 total IgG
The rate of specific total IgG obtained in this age group are significantly different between groups of hemoglobin. Thus the rate of total IgG against the antigenic peptides at subjects with hemoglobin E carrier is higher than those of hemoglobin A (Figures 4-6). This difference was significant (p=0.004). There is therefore a different acquisition of specific total IgG in subjects older than 20 years with hemoglobin E carrier.
In our study, we have observed that the rate of hemoglobin E is (58%). These results are higher than those found by Fucharoen G et al. [20] in Thailand (50%) and Flatz, et al. [21] Laos (43%).
Our data nonetheless confirm the strong representation of hemoglobinopathies E in Asia and its coincidence with the current distribution of malaria. Hemoglobin S is found in sub-Saharan Africa, in the equatorial belt, the Middle East and parts of India. The Hemoglobin C is common in the northwest of Africa where its distribution coincides with that of hemoglobin S. They are absent in Asia.
We also observed that the rate of specific IgG against MSP-1 levels is significantly higher than SR-11.1 and MSP-2 in the age groups. We have noted that the recognition of specific antibodies to the three peptides MSP-1, MSP-2 and SR-11.1 for the individuals in our study is low.
These low titers of antibodies specific for these antigens make it difficult to evaluate a clear relationship in the different groups of hemoglobin. This result could be explained by the variability of response options to many antigens of parasite strains. This could also be explained by the fact that IgG2 (and IgG4) are considered non cytophilic antibodies capable of blocking the protective mechanisms by preventing the recognition of antigens by antibodies cytophilic [22,23].
Three antigenic peptides we had used, MSP-1 and MSP-2 appear much more immunogenic than SR-11.1.
In our study, no significant differences between the titers of specific total IgG in hemoglobin groups have been found in subjects less than 20 years. Ntoumi et al. [15] in 2005 in Gabonese children with hemoglobin AS showed a difference in antibody titers. This could be explained by the complex pathways of invasion of red blood cells by P. falciparum which increase the titers of IgG cytophilic against MSP-1 and MSP-2.
Our results could be explained by the fact that in endemic areas, the acquisition of antimalarial immunity required among young people depends on the level of transmission and is age-dependent [24,25]. In our population, subjects under 20 years have not yet reached the peak maximum amount of specific IgG.
We observed in the age group of over 20 years that the specific total IgG levels of antigenic peptides (MSP-1, MSP-2 and SR-11.1) are higher among carriers of hemoglobin E that hemoglobin AA carriers. This difference is significant. The same was done in Burkina Faso by Verra et al. [26]. in 2007 which showed that the specific total IgG levels against the malarial antigens (MSP-1, MSP-2, MSP-3, EBA-175, and AMA-1) were higher when the subject was hemoglobin S or C carrier.
These results suggest that hemoglobin E carriers have better production of specific total IgG MSP-1; MSP-2 and SR-11 epitopes compared to normal hemoglobin AA carriers. It is possible that the multiplicity of P. falciparum genotypes contributes to increasing the recognition repertoire of P. falciparum and thus to accelerate the acquisition of protective immunity against the parasite.
The analysis of our results shows that the frequency of hemoglobinopathies E (58%) higher in Vietnam. As we have shown that the response of specific total IgG against the plasmodium antigens (MSP-1, MSP-2 and SR-11.1) is improved in patients over 20 years with hemoglobin E genotype compared to those with the AA genotype. In subjects under 20 years, no significant differences were obtained between the different groups of hemoglobin. Even if we got a low rate of IgG against the antigenic peptides MSP-2 and MSP-1 appear much more immunogenic than the SR11.1 in different age groups.
Conceived and designed the experiments: SY TY FF. Performed the experiments: SY TY FF. Analyzed the data: SY TY.FF Contributed reagents/materials/analysis tools: SY TY FF.SI BW Wrote the paper: SY FF TY SI SG.
We declare that we have no conflict of interest.
Download Provisional PDF Here
Article Type: Research Article
Citation: Sourabié Y, Bazié WW, Sangaré I, Sanou G, Fumoux F, et al. (2016) Immunological analysis of Malaria infection in carriers of abnormal Hemoglobin EA or EE. Int J Vaccine Immunizat 2(2): doi http:// dx.doi.org/10.16966/2470-9948.110
Copyright: © 2016 Sourabié Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access